Application of Captisol® Technology to Enable the Formulation of Remdesivir in Treating COVID-19
How Captisol works
James Pipkin, PhD, Vince Antle, PhD, and Rebecca Garcia-Fandiño, PhD, explain why more Captisol® than ever is required to meet Gilead’s bold goals of making remdesivir available to COVID-19 patients in the US and to hundreds of countries around the world. Check the full article on drug-dev.com
Captisol® is a polyanionic beta-cyclodextrin derivative with a sodium sulfonate salt separated from the lipophilic cavity by a butyl ether spacer group, or sulfobutylether (SBE). Captisol® is not a single chemical species, but comprised of a multitude of polymeric structures of varying degrees of substitution and positional/regional isomers dictated and controlled to a uniform pattern by a patented manufacturing process consistently practiced and improved to control impurities.
The selection of Captisol® and its desirable safety profile and drug solubilization properties was based upon extensive evaluations of the mono, tetra and hepta-substituted dominated compositions. Captisol® is the trade name for Ligand’s modified beta-cyclodextrin technology.
Several hundred of pre-clinical and clinical studies have been performed and indicate that Captisol® is safe when administered parenterally or orally and does not exhibit the nephrotoxicity associated with beta-cyclodextrin. Relative to beta-cyclodextrin, Captisol® provides higher interaction characteristics and superior water solubility in excess of 100 grams/100 ml – a 50-fold improvement.
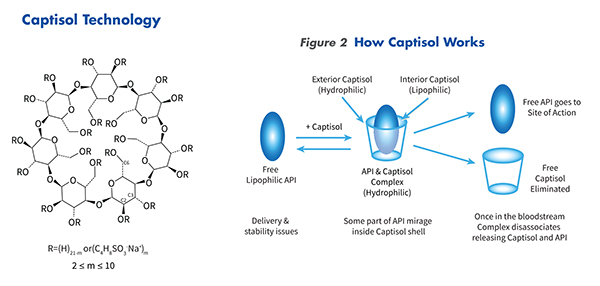
How does Captisol® Work
Traditional formulation systems for very insoluble and/or unstable active pharmaceutical ingredients (APIs) have involved a combination of organic solvents, surfactants and extreme pH conditions. These formulations may precipitate upon injection, or may cause irritation and adverse reactions. At times, these approaches are inadequate for solubilizing enough active agent for a preferred formulation.
Captisol® Solubilizes
Neutral, cationic and anionic APIs have been effectively associated with Captisol®. Aqueous solubilities have increased by a factor of 10 to 25,000, depending on the compound. In contrast to other solubilization technologies, product or traditional formulation system, the feasibility and solubility effectiveness of Captisol® can be rapidly assessed with a few simple lab experiments. More information on Captisol®









































 All4Nutra
All4Nutra







