3D printing technology on the verge to create booming growth in pharmaceuticals
3D printing technology on the verge to create booming growth in pharmaceuticals
1. Pharmaceutical 3D printing
Recent advancements in the area of 3D printing (3DP) have prompted scientists to test and evaluate the technology’s applicability in the medical sector. Drug delivery systems, tissue engineering, tissue and organ models, prosthetics and replica manufacturing, implants, and many more fields are among those that are rapidly developing (1). As a result, 3D printing is very important in the field of pharmacology. Its use in this field serves as the foundation for a new patient-centered therapy approach. Personalized medications and treatments may be created via additive pill manufacturing. This may assist patients in receiving the most effective therapy at the lowest possible cost. Scientists have been actively investigating the possibilities of 3D printing in the pharmaceutical sector over the last five years (2).
Three-dimensional printing is a method in which digitally controlled 3D shapes are created by layer-by-layer deposition of materials. It was formerly extensively utilized for fast prototyping in the automotive, robotics, aerospace, and other sectors. The use of 3D printers in pharmaceutical research has grown dramatically as a result of the broad availability of commercial 3D printers and the recent FDA clearance of the first 3D printed dosage form (Spritam) (3). The ability to produce complicated, customizable and personalized-on-demand dose forms is the primary reason for researchers’ increasing interest in 3D printing. Customized vaginal progesterone rings, channelled tablets for better disintegration and dissolution, personalized oral delivery devices, and a topical nose-shaped device for the treatment of acne have all recently been created using fused deposition modeling (FDM) 3D printing.

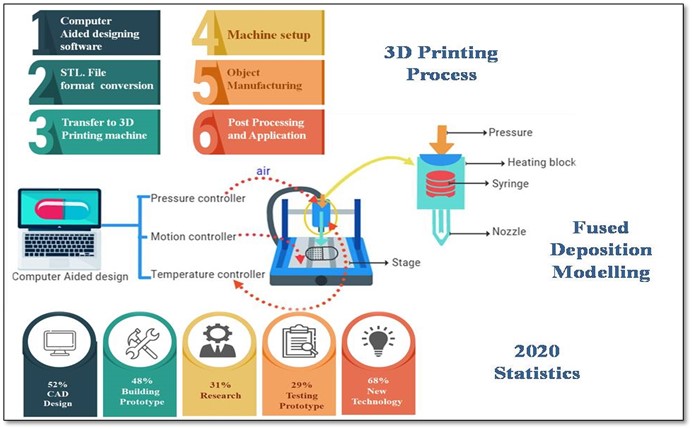
2. Process of designing pharmaceuticals
2.1 Computer-aided designing software
The first stage in the 3D printing process is to create a digital model. Computer-Aided Design is the most popular technique for creating a digital model (CAD). Through 3D scanning, reverse engineering may also be utilized to create a digital model. CAD modeling and 3D scanning are also available. When designing for 3D printing, there are many design issues to consider. These are usually concerned with feature geometry restrictions, support material, and the need for an escape hole.
2.2 Conversion of file and transferring to machine acceptable format
A CAD model must be translated into a format that a 3D printer can understand to 3D print a component. The first step is to convert the CAD model into a Stereolithography (STL) file, which is also known as a Standard Triangle Language file. 3D printing file formats such as OBJ and 3DP are also allowed, although they are less popular (4). STL describes an object’s surfaces using triangles (polygons), thus reducing the sometimes complicated CAD representation. The majority of CAD applications can export a drawing as an STL file. After creating an STL file, it is loaded into slicer software, which slices the design into layers that will be utilized to construct the component. The STL file is converted to G-code by the slicer software. G-code is a numerically controlled software application that is used in computer-aided manufacturing (CAM) to operate automated equipment such machines and 3D printers. The slicer software also enables the 3D printer operator to select support location, layer height, and component orientation to define the 3D printer build parameters. Although certain universal slicer applications exist, such as Netfabb, Simplify3D, and Slic3r, slicer programs are typically unique to each type of 3D printer. As a designer, you often just need to submit an STL file to a 3D printer operator (5). The operator will then configure the appropriate print settings and generate the G-code file on their own.
2.3 Machine setup and printing characteristics
To produce an exact print, most Fused Deposition Machine (FDM) machines include a number of settings that may be modified. The operator controls the uniformity of the extruded filament by adjusting the build speed, extrusion speed, and nozzle temperature (some machines use automatic presets based on the type of material that is being printed). The resolution of an FDM printed item is defined at the most basic level by the nozzle diameter and layer height. While all parameters affect a part’s dimensional accuracy, a smaller nozzle diameter and lower layer height are often recommended for components that need a smooth surface and high degree of detail. When printing with FDM, the available build volume must be taken into account. Desktop printers typically have a build chamber that is 200 x 200 x 200 mm. Build chambers as big as 1000 x 1000 x 1000 mm is available on larger industrial equipment. Breaking down a design into components that can be combined after printing is frequently the best option for extremely big items.
2.3 Object manufacturing and post processing
During object manufacturing, there are several parameters to be considered such as the geometry of an object, infill parameters and support structure if the shape needs any support while printing (6). “Bi-layer or multi-layer may give more than one medication in one dosage, which is more convenient. By delivering the drug in different layers, incompatibilities between the drugs can be avoided that would otherwise be impossible to avoid in a single monolithic structure. Multiple layers also enable various speeds of distribution of the drug, and multi-layer tablets help to establish brand identification (7). We can even design tablets with different release patterns just by changing their inner geometry by using the fused deposition 3D Printing technique figure 2. Priming and cleaning are the most common methods of post-processing.

3. Fused deposition modeling
The thermoplastic feedstock filament is pulled from a spool into the FDM liquefier head by drive wheels, where it is heated to a semi liquid condition before being extruded out as ultrathin beads via the aperture of a nozzle tip placed at the bottom of the head. When required, a feedstock filament of support material is pulled into the liquefier head from another spool and deposited via another nozzle tip (8). The fundamental FDM method is shown in Figure 1, and a liquefier head with the model material flow is depicted in Figure 2. The computer controls the movement of the head, which distributes the material in tiny layers on a fixtureless base. The component is built layer by layer, with the material solidifying and bonding to the preceding layer as soon as it is deposited from the nozzle tip. To assist the construction process, the build chamber may be adjusted to a temperature lower than the melting point of the extruding material. After that, the finished component is ready to be taken from the FDM machine. Later, support structures are broken away from the component or the whole model is dipped in a solution (9). The process settings determine the part’s accuracy and precision, and some high-end FDM machines provide very high precision.
4. 2020 Statistics of 3D Printing
Many people from different sectors and nations participated in the 2020 research. This research yielded some intriguing data. According to the research, the most common industrial use is Building Prototype 48 %, CAD 52 %, Research accounted for 31% of the total. Prototype testing 29% (10). In addition, 68 percent of survey participants indicated an interest in learning more about innovative 3dp uses in technology. The 3D printing business is constantly changing and developing, making it tough to stay up with the latest developments. Keeping track of 3D printing data is an excellent way to remain informed. In the meantime, 3D printing usage is unequal across geographical areas. North America and Europe are now leading the push. However, the two areas are in danger of losing their leadership positions in the global 3D printing sector, with Asia quickly emerging as a powerful competitor. The orthopedics 3D printing market was valued at $691 million in 2018 and is expected to rise to $3.7 billion by 2027, according to SmarTech Analysis.
Find out more about 3D printing here!
Article information: Prachi Khamkar. Project Mentor, Pharmaceutical Manufacturing Operations, CiRee, Pune.
References:
- Please cite this article as: Al-Dulimi Z, Wallis M, Tan DK, Maniruzzaman M, Nokhodchi A, 3D printing technology as innovative solutions for biomedical applications, Drug Discovery Today (2020), doi: https://doi.org/10.1016/j.drudis.2020.11.013
- https://www.europeanpharmaceuticalreview.com/article/119865/the-uses-and-prospects-for-3d-printing-in-pharmacology/
- Aprecia Pharm (2020) Spritam® (Levetiracetam), Aprecia Pharm
- Yeong, W. et al. Indirect fabrication of collagen scaffold based on inkjet printing technique. Rapid Prototyp J (2006) 12, 229–237
- Rayate, A. and Jain, P.K. A review on 4D printing material composites and their applications. Mater Today Proc (2018) 5, 20474–20484
- Reddy Dumpa, N., Bandari, S., & A. Repka, M. (2020). Novel Gastroretentive Floating Pulsatile Drug Delivery System Produced via Hot-Melt Extrusion and Fused Deposition Modeling 3D Printing. Pharmaceutics, 12(1), 52. MDPI AG. Retrieved from http://dx.doi.org/10.3390/pharmaceutics12010052
- Ghanizadeh Tabriz, A., Nandi, U., Hurt, A., Hui, H., Karki, S., & Gong, Y. et al. (2021). 3D printed bilayer tablet with dual controlled drug release for tuberculosis treatment. International Journal Of Pharmaceutics, 593, 120147. doi: 10.1016/j.ijpharm.2020.120147
- Goyanes, A., Buanz, A., Basit, A., & Gaisford, S. (2014). Fused-filament 3D printing (3DP) for fabrication of tablets. International Journal Of Pharmaceutics, 476(1-2), 88-92. doi: 10.1016/j.ijpharm.2014.09.044
- Tips for Quick, Thorough Soluble Support Removal. (2021). Retrieved 20 July 2021, from https://www.stratasys.co.in/explore/blog/2012/tips-for-quick-thorough-soluble-support-removal
- A Guide to 3D Printing | An introduction and overview to the industry. (2021). Retrieved 20 July 2021, from https://planetechusa.com/a-guide-to-3d-printing/









































 All4Nutra
All4Nutra







