A novel test system for the evaluation of oral mucoadhesion of fast disintegrating tablets
A novel test system for the evaluation of oral mucoadhesion of fast disintegrating tablets
11. September 2018
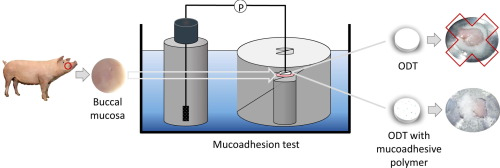
Oromucosal drug delivery is necessary when a local effect in the oral cavity is required. Bioadhesive formulations should be advantageous because a larger fraction of the active principal is retained at the site of action allowing for an enhanced and prolonged effect. Despite a variety of mucoadhesion test systems being described in literature, none of these in-vitro tests does relate to physiological conditions in the oral cavity and suites for the testing of complete dosage forms, e.g. tablets. The novel mucoadhesion test is based on porcine buccal mucosa.

Constituents, osmolality and pH of the used artificial saliva was as close as possible to physiologic conditions and the flushing rate was equivalent to the physiological secretion rate of saliva. Suitability of the novel test was evaluated with ODTs with live probiotic bacteria as the active principle. With the novel test system, it was shown that in the presence of mucoadhesive polymers (Carbopol®, Metolose® and chitosan) up to 30 % of the probiotic bacteria adhered to the mucosa corresponding to a two‐ or threefold increase compared to tablets without such polymers. Furthermore, the influence of the tablet size (contact area) on the mucoadhesive properties could be resolved.









































 All4Nutra
All4Nutra







