1. Introduction
The primary goal of any dosage form is to ensure the accurate and efficient release of a specific amount of drug, resulting in the desired pharmacological response [1].
Hereby, the USP recognizes two main categories of drug release: the immediate release and the modified release. The latter has two further classifications: the delayed release and the extended release [2]. In the context of oral products, such terms as “enteric-coated” and “gastro-resistant” describe the prevention of drug release in the stomach while facilitating it in the intestine [2].
A delayed drug release is typically achieved via enteric coating of dosage forms such as tablets, capsules, and subparts [3]. The primary purpose of enteric coating is to protect the gastric mucosa from potentially irritating drug compounds or to prevent the degradation of the active pharmaceutical ingredient (API) due to acidic conditions or stomach enzymes [4].
The majority of film-forming polymers used in enteric coatings are weak acids containing carboxylic acid groups. At low pH, these groups undergo protonation, leaving the polymer non-ionized and insoluble in gastric acid [5]. However, as the pH increases in the small intestine, the carboxylic groups become ionized, resulting in the solubility of the polymer [5], leading to the release of the API. The deprotonation and solubility characteristics of a polymer depend on such factors as the pKa of the acidic groups, the quantity of free carboxylic groups in the polymer chain, and the pH of the surrounding environment. Modifying these properties of the polymer makes it possible to precisely control the release behavior of enteric-coated tablets at specific locations in the small intestine or the colon [6].
Enterically-coated tablets and delayed-release capsules are known for their effective functions. However, this enhanced functionality also adds complexity and cost to the manufacturing process, resulting in higher prices for the drug product. Hence, Evonik has presented as functional pre-locked enteric-protected capsules, denoted as EUDRACAP® enteric (Figure 1). The capsules combine the well-established EUDRAGIT® polymer coating with standard empty pre-locked HPMC capsules [7]. It was specifically designed to accommodate a variety of drug forms, including powders, pellets, granules, and selected liquids. One of the key advantages of these capsules is the easy opening and closing mechanism, which integrates seamlessly with standard capsule filling systems [7].
EUDRACAP® enteric offers robust acid resistance for up to 4 hours without the use of capsule banding [7], ensuring the protection of the encapsulated medication. This concerns the details outlined in the monograph of the Ph.Eur. for capsules [8], where the features of gastro-resistant capsules as delayed-release capsules are described that are intended to resist the gastric fluid and to release their active substance or substances in the intestinal fluid. Additionally, these capsules are designed to be precise in terms of pH targeting, enabling the desired drug release profile. These attributes offer sufficient control of drug delivery. According to the manufacturer, there are additional advantages of this particular product:
Firstly, the capsules can be manually filled, eliminating the need for specialized equipment and the need for capsule banding [7]. This feature not only simplifies the production process but also opens up possibilities of manufacturing delayed-release dosage forms locally, e.g., in pharmacies. Secondly, the capsules allow direct filling with powder, eliminating the use of coated pellets. This not only streamlines the manufacturing process but also makes it more accessible and cost-effective for consumers.
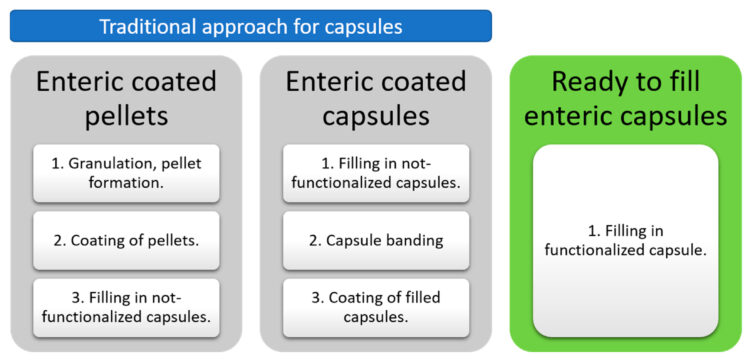
The use of pre-locked enteric-protected capsules presents a compelling advantage in terms of manufacturing efficiency in comparison with the traditional approaches (Scheme 1).
The cases of enteric-coated pellets and enteric-coated capsules demand at least three unit operations, which also imply a meticulous optimization of various process parameters, which can be time- and resource-consuming [9]. However, with a ready-to-fill enteric-protected capsule, the manufacturing process is notably simplified. With only one manufacturing step involved, the need for extensive process development and optimization is significantly reduced. This not only simplifies the production process but also makes it suitable for heat and moisture-sensitive APIs, enhancing versatility and efficiency in pharmaceutical manufacturing. This translates to significant cost savings and acceleration of the drug development time.
In this paper, the aim was to validate the claims by the manufacturer and to test if indeed the in vitro performance of
EUDRACAP® enteric is superior to the existing commercial finished drug products. In order to do so, we carried out a test using two representative drugs that require enteric protection.
Diclofenac is an example of such a drug with gastric irritant properties [10]. It is a widely used nonsteroidal anti-inflammatory drug for the treatment of chronic arthritis and mild-to-moderate acute pain [11]. It has a short plasma half-life of approximately one to two hours [11]. Various formulations of diclofenac are used for the drug products available in the market or described in the literature [12,13,14,15], including enteric-coated tablets and encapsulated enteric-coated pellets. In this particular case, the capsules are only applied for easier administration and do not have any protective function. As a result, the production is significantly more expensive, because it requires an additional pellet production and coating step before encapsulation. In addition, the protective function of the enteric coating may theoretically decrease, as the surface area exposed to the acidic environment increases.
Also, acid-sensitive drugs, such as proton pump inhibitors, require enteric protection from gastric acid [16]. Omeprazole, a common proton pump inhibitor used for the treatment of gastric acid secretion-related disorders, was introduced in Europe in 1988 and in the United States in the 1990s [17]. Since its introduction, it has played a significant role in the management of patients requiring antisecretory drugs. Omeprazole is known to degrade in acidic environments, depending on the pH levels, while exhibiting acceptable stability under alkaline conditions. Therefore, omeprazole can serve as an example of modified-release delivery [18].
The degradation process of omeprazole is easily detectable since it occurs rapidly and is visually noticeable due to the bright yellow color of the degradation products. Additionally, most of the impurities generated during its degradation are extensively documented in the European and US pharmacopeia [19,20]. In addition, the market offers a wide variety of omeprazole formulations, with enteric-coated tablets and encapsulated pellets being among the most prevalent choices [21,22].
In the light above, we selected diclofenac and omeprazole for the evaluation of the enteric protection provided by Evonik’s EUDRACAP® enteric capsules. This evaluation included the assessment of related substances after acidic exposure according to the USP <711> and Ph.Eur. 2.9.3 for delayed-release formulations [23,24] by high-performance liquid chromatography (HPLC), with the goal of comparing their content to an existing marketed finished drug product.









































 All4Nutra
All4Nutra







