A large number of dietary supplements are available in solid oral dosage forms. Vitamin and mineral supplements can be easily pressed into tablets by wet granulation or direct compression, but such tablet formulations require good binders. Commonly used binders in tablet formulations include cellulosic materials, such as hydroxypropyl cellulose (HPC) or microcrystalline cellulose (MCC), or synthetic polymers, such as polyvinyl pyrrolidone (PVP). For organically oriented consumers, however, these excipients sound synthetic and unnatural.
The trend toward organic foods has been evident for several years as consumers develop a greater awareness of organic products [1]. Dietary supplement manufacturers are also slowly changing to organic formulations, as consumers increasingly look to avoid chemical or synthetic ingredients. Therefore, dietary supplement manufacturers are under increasing pressure to replace traditional bind-ers with more natural-sounding ingredients.
The same challenge applies to fillers, flow agents, lubricants, disintegrants, and pigments, including magnesium stearate, stearic acid, silica, and synthetic colors. This move toward more natural ingredients is part of the so-called “Clean Label” movement, which also attaches importance to the sustainability of raw materials.
Pharmaceutical manufacturers are also beginning to use more natural ingredients as excipients due to their increasing availability, relatively low costs, lower toxicity, and fewer side effects. The application of natural ingredients in pharmaceuticals is expected to expand in the coming years along with consumer demand. The aging population in developed countries is expected to increase the future rate of chronic disease and stimulate demand for pharmaceuticals. At the same time, consumers are looking to natural products to improve their general well-being and prevent illness [2].
Download the whole article as a PDF here
By working closely with their ingredient suppliers, tablet manufacturers can successfully change a formulation from a synthetic binder to natural or organic ingredients.
Replacing synthetic binders
Binders give cohesion to the loose particles or granules in a tablet formulation. This ensures that the tablet remains intact after compression. To replace conventional binders such as HPC, MCC, and PVP, a natural binder must be able to produce tablets with the same mechanical strength and release properties. Table 1 shows some starches, gums, mucilages, and dried fruits that have binding capacity as well as other characteristics, such as filler and disintegrant properties, and that could be used as alternatives to synthetic binders.
By working closely with their ingredient suppliers, tablet manufacturers can successfully change a formulation from a synthetic binder to natural or organic ingredients. A natural ingredient cannot replace a synthetic ingredient 1:1, however. Users usually must add several natural raw materials in different concentrations to a formulation to achieve the desired binding properties. Customer-specific premixes made from various certified natural raw materials, on the other hand, can simplify development, handling, and the manufacturing process.
Natural disintegrants
Disintegrants are substances or mixtures of substances formulators add to a formulation to increase the dispersion or breakup of tablets and capsules into smaller particles for fast dissolution. Popular synthetic superdisintegrants include crosslinked PVP, MCC, croscarmellose sodium, and sodium starch glycolate, all of which are chemical-sounding names that are unsuitable for clean label products. More natural sounding raw materials that can act as disintegrants include: mucilage of ispaghula or psyllium husks, cress, gum karaya, fenugreek seed and gum of locust bean, chitin and chitosan, gellan, agar, alginates, oat fiber, xanthan, cucurbita maxima pulp powder, hibiscus rosasinensis Linn, and mango peel pectin.
As with dry binders, 1:1 disintegrant replacement is not possible in most cases. A dry powdered premix compound based on natural and certified organic ingredients (such as Biogrund’s CompactCel® DIS) can replace synthetic disintegrants in tablet, capsule, and granule formulations and enable the rapid breakup upon contact with moisture required by a fast-release solid oral dosage form.
From oil to powder to capsules naturally
Manufacturers are increasingly looking to add natural sources of vitamins, probiotics, omega 3-6-9 fatty acids, antioxidants, amino acids, and hemp to their natural supplement products. These nutrients often come in the form of an oil or oily powder. For oily powders, micro-encapsulation can be beneficial, especially if the oily powder is then filled into hard capsules, which allows precision dosing of ingredients or nutrients. Flavor and odor masking can minimize unpleasant tastes and smells associated with certain nutrients. Protection from moisture, acids, heat, and oxygen is also critical, as it enhances stability, bioavailability, and delivery. The powder must also be easy to handle and free flowing.
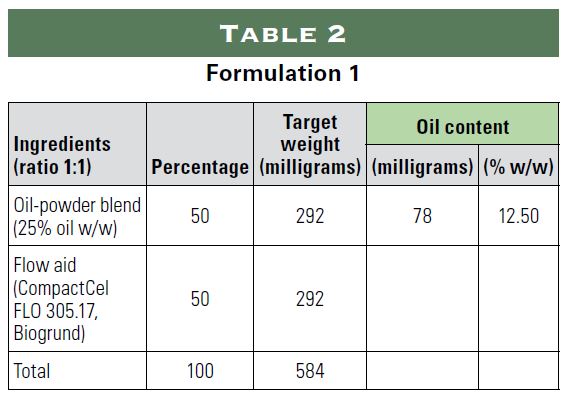
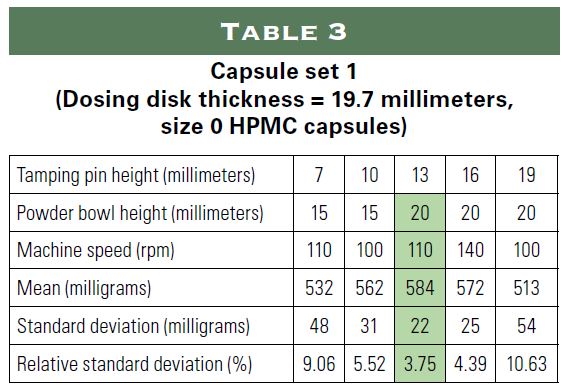
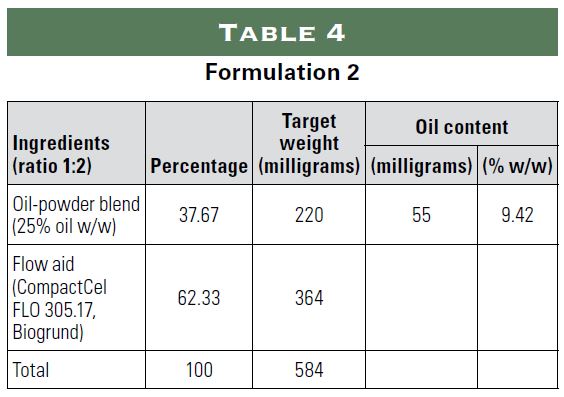
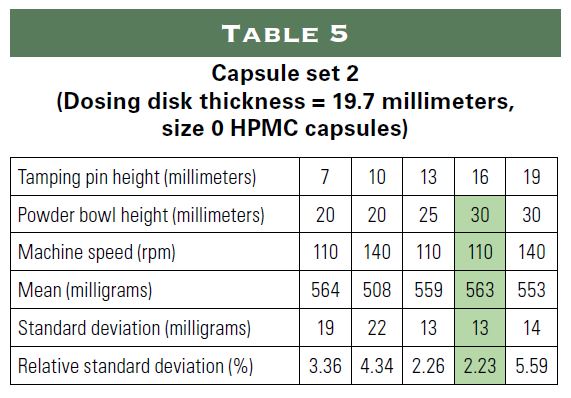
The following study examined the possibility of incorporating oily substances into a powder for encapsulation using only natural ingredients. The study simulated the following three applications using sunflower oil as an oil substitute:
1) 20 milligrams of hemp in a size 0 capsule;
2) 22.5 international units (IU) of vitamin E and approximately 15 milligrams of vitamin D3 in a size 1 capsule; and
3) 30 million colony forming units (CFU) of probiotics in a size 0 capsule using oil to make the probiotics more stable.









































 All4Nutra
All4Nutra