This article presents a novel 3D printing technique called L-FDM (liquid for fused deposition modeling), which is based on the deposition of molten thermoplastic material. The new method allows for the direct introduction of chemicals and polymer filament modifications during the printing process. In contrast to traditional incremental methods, L-FDM eliminates the need for extra granulating, extrusion, and processing equipment, making it possible to introduce chemical additives to the polymer matrix directly. This opens up exciting possibilities for chemical laboratories to test and experiment with new and known chemicals through 3D printing. The article discusses the technical aspects of L-FDM and its potential applications and provides practical examples of direct filament modifications using the technique. The results of these modifications were verified using a colorimeter, electron microscopy (SEM/EDS), and optical microscopy.
1. Introduction
1.1. State-of-the-Art
Since the 1980s, there has been a rise in contemporary additive manufacturing (AM) technologies, commonly referred to as 3D printing. These technologies enable the use of a wide range of materials, including polymers, metals, ceramics, food, and biomaterials [1]. Three-dimensional printing provides several benefits, such as the ability to produce objects directly without the need for a complete technological infrastructure, the capacity to manufacture products with complex geometries, design flexibility, cost-effective use of materials, and eco-friendliness [2]. Among the various AM techniques, the most significant methods are stereolithography (SLA), selective laser sintering (SLS), and fused deposition modeling (FDM). Stereolithography based on photopolymerization was the first method developed by additives, using UV-cured resins as the building material [3].
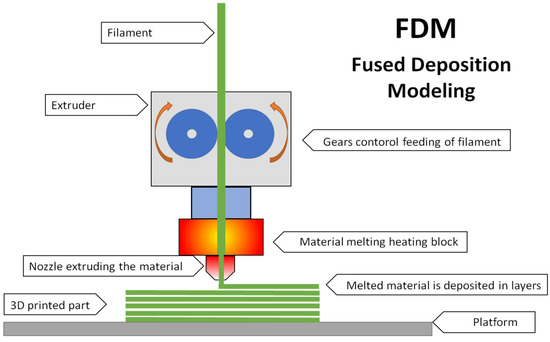
Compared to other methods, this technique is known for its high accuracy in producing objects without post-processing. The SLS method is a printing technology based on a powder polymer. In this method, the laser beam moves along a specific path and sinters the polymer powder under the influence of the generated heat in layers until reaching a three-dimensional model [4]. Fused deposition modeling or fused filament fabrication (FDM/FFF) was developed in 1988 by Scot Crump, founder of Stratasys, and marketed under the name FDM (US Patent No. 5 738 817). In the process, we use a thermoplastic filament as the building material, which is mounted on a spool and fed to the head, where it reaches the appropriate melting temperature. Printing consists of depositing molten material through a nozzle of a certain diameter layer by layer until a three-dimensional object of the desired shape and geometry is reached [5,6]. FDM technology using thermoplastic filament is the most preferred in industrial applications. The FDM method is the most preferred of all those mentioned due to the low cost of devices (3D printers) and easy availability, low price, and variety of filaments. From a technical perspective, 3D printing technology is a very important conceptual element of the fourth industrial revolution or Industry 4.0. It brings many benefits, such as digital data transfer, remote access, minimal human intervention, the ability to develop complex geometries and intelligent materials, less waste generation, and lower final processing requirements. All these features fit perfectly into the goals of Industry 4.0 [7]. Furthermore, they meet the growing market requirements, which include rapidly changing customer requirements, a demand for high-quality products at the lowest possible production costs, especially in low-volume production scenarios, and the production of products for the needs of individual entities [2].
Although 3D printers are becoming more common in households, their potential use in scientific laboratories is often underestimated by researchers, and their application as laboratory equipment is not considered. These devices can be very useful in many different types of laboratories, from chemical to biological, pharmaceutical, and materials science [8,9,10]. The use of 3D printing is becoming particularly popular in tissue engineering for medical purposes [11]. The scientific field that has particularly benefited from the emergence of this technology and the possibility of rapid prototyping directly in the laboratory is undoubtedly microfluidics. In the past, the production of a microfluidic device was a troublesome task involving many stages. This process has changed with the emergence of 3D printing, which has enabled the execution and printing of various projects within just a few hours, accelerating research and reducing prototyping costs [8,12]. Using additive techniques, it is possible to design vessels for chemical reactions, thanks to the ability to add substances and additional components, such as filters or adsorbents (e.g., silica gel), during the printing process. The versatility of printing and the ability to add external components to the manufactured object allows for precise and repeatable stages of synthesis and purification [8,13]. Three-dimensional printing has also been applied in pharmacies as a tool for obtaining personalized tablets with customized drug release profiles [8,14]. Another field that has extensively utilized additive manufacturing techniques is catalysis. Additive printing devices, in this case, can be used to produce flow reactors and static mixers [10,15].
1.2. Liquid for the Fused Deposition Modeling Technique (L-FDM)
The conducted research aims to develop an innovative, unconventional method of introducing chemical substances into the polymer matrix of materials dedicated to 3D printing. This method can be easily carried out in any chemical laboratory without the need for conventional plastic processing equipment (Figure 1).
This research is based on using a 3D printer as a laboratory tool to introduce various chemical substances, e.g., dyes, solvents, organic compounds, and organosilicon compounds, into a polymer matrix. Most studies on polymer modification rely on traditional techniques that involve adding additives during the liquid polymer state. These methods include extrusion, mixing, rolling, and kneading in mixers [16] or mixing the modifier with a polymer solution in a volatile solvent and the subsequent removal [8]. This article discusses methods that eliminate the need for costly and energy-consuming processing equipment. These methods can be utilized in any laboratory by users without access to specialized devices. The proposed method of introducing chemical substances into the polymer matrix consists of surface modification of the filament through immersing in a solution (see Figure 3).
The filament is passed through a reservoir containing a modifier (a liquid chemical substance or its solution) and then through a drying or excess substance removal system. During the next stage, the filament that has been coated with the modifier is fed directly to the 3D printer head. Once there, it is melted and extruded through a nozzle, forming a three-dimensional object layer by layer. This method enables the testing of a large number of substances with high efficiency and throughput for creating previously unknown functional materials that open up entirely new application possibilities. Currently, tens of millions of different chemical compounds are discovered worldwide, and the properties and potential uses/applications of most of them have not yet been explored. Most additives introduced into the polymer matrix serve functional purposes, such as improving product durability under different atmospheric conditions or enhancing antistatic properties, increasing the strength and usability, changing the thermal properties, and modifying the color, among others [17,18,19,20].
Coating a filament’s surface with a liquid modifier can occur either during printing (Figure 4(2.1,2.2)) or separately outside the printing process (Figure 4(2.3,2.4)). The L-FDM printing technique enables the use of one reservoir with a liquid modifier (Figure 4(2.1)) or multiple reservoirs with different chemical substances capable of dissolving in various solvents (Figure 4(2.2)). The filament modification presented in the diagrams in Figure 4(2.3,2.4) also involves passing the filament through a reservoir containing a modifier and a drying system, with the difference that, in the next stage, it does not go directly to the print head but is rewound onto the spool. This method also allows for multiple coatings of the filament surface with a chemical substance or the use of several different substances that dissolve in different solvents by winding the modified filament back onto the spool and passing it through the system again (Figure 4(2.3)) or by using multiple reservoirs with different modifiers (Figure 4(2.4)). This feature enables the filament to be utilized at a different location or time than the modification procedure itself.
In this article, we demonstrate the feasibility of incorporating particular substances into the polymer matrix during the printing process through the use of L-FDM technology. The obtained modifications were validated by a colorimetric analysis, optical microscopy, and scanning electron microscopy with energy-dispersive X-ray spectroscopy (SEM/EDS).
Download the full study as PDF here: Liquid for Fused Deposition Modeling Technique (L-FDM)—A Revolution in Application Chemicals to 3D Printing Technology: Color and Elements
or read it here
Przekop, R.E.; Gabriel, E.; Pakuła, D.; Sztorch, B. Liquid for Fused Deposition Modeling Technique (L-FDM)—A Revolution in Application Chemicals to 3D Printing Technology: Color and Elements. Appl. Sci.2023, 13, 7393.
https://doi.org/10.3390/app13137393










































 All4Nutra
All4Nutra