Devices to overcome the buccal mucosal barrier to administer therapeutic peptides
Devices to overcome the buccal mucosal barrier to administer therapeutic peptides
Abstract
Peptide therapeutics are important in healthcare owing to their high target specificity, therapeutic efficacy, and relatively low side effect profile. Injections of these agents have improved the treatment of chronic diseases including autoimmune, metabolic disorders, and cancer. However, their administration via injections can prove a barrier to patient acceptability of treatments. While oral delivery of these molecules is preferable, oral peptide formulations are associated with limited bioavailability due to degradation in the intestine and low epithelial permeability. Buccal administration of peptides is a potential alternative to injections and oral formulations. Similar to the oral route, the buccal route can promote better patient adherence to dosing regimens, along with the added advantages of not requiring restriction on food or drink consumption before and after administration, as well as avoidance of the liver first-pass metabolism. However, like oral, effective buccal absorption of peptides is still challenging due to the high epithelial permeability barrier. We present a multidisciplinary approach to understanding the buccal physiological barrier to macromolecule permeation and discuss how engineered devices may overcome it. Selected examples of buccal devices can facilitate fast and efficient macromolecule absorption through multiple mechanisms including physical disruption of epithelia, convection-based mass transfer, and a combination of physicochemical strategies. Importantly, minimally invasive devices can be self-applied and are associated with the maintenance of the barrier after exposure. We analysed the critical attributes that are required for the clinical translation of buccal peptide administration devices. These include performance-driven device development, manufacturing features, patient acceptability, and commercial viability.
Introduction
Therapeutic use of peptides and proteins has increased since the inception of recombinant protein manufacturing technology in the 1980 s [1], [2]. The era of peptide therapeutics commenced with the discovery of insulin in 1922, which established it for treatment of Type 1 diabetes (T1D). Peptide and protein therapeutics find numerous applications in alleviating diabetes, obesity, cancer, metabolic dysregulation, endocrine disorders, prevention of transplant rejection, and as components of vaccines [2], [3], [4], [5] (Fig. 1). Peptide and protein therapeutics perform complex biological functions due to structures which promote specific high-affinity target binding. This is unlike small molecules, whose less specific binding can increase the risk of off-target effects. Peptides, the focus of this review, comprise short chains of amino acids, typically ranging from 2 to 50 amino acids, while proteins are larger and more structurally complex. Strict definitions vary and, for example, insulin can be described as either a peptide or a protein, and we use the former definition here. Peptide and protein therapeutics can be classified into Group 1: Peptide and protein therapeutics with enzymatic or regulatory activity; Group 2: Peptide and protein therapeutics with targeting activity; and Group 3: Protein vaccines [2]. Examples of Group 1 agents include Glucagon-like Peptide 1 (GLP-1) analogues, insulin, growth hormone, and erythropoietin; Group 2 covers humanized monoclonal antibodies (mAbs) and antibody-drug conjugates (ADCs), while Group 3 includes protein vaccines (e.g. vaccines for hepatitis B and HPV) [1], [2], [6] (Fig. 1). Additionally, chemically modified proteins and fusion proteins are a major therapeutic modality [6].

Fig. 1. Clinical applications of peptide and protein therapeutics.
Monoclonal antibodies (mAbs) and antibody-drug conjugates (ADC) are also included (shaded) since they are protein-based.
Abbreviations: ALL = Acute lymphoblastic leukaemia; Fam-Trastu = Fam-Trastuzumab deruxtecan-nxki; Inotu-ozo = Inotuzumab ozogamicin; Belanta-maf = Belantamab- mafodotin.
Currently, the global market share of peptide and protein therapeutics is ∼ US $ 400 billion, with more than 350 FDA-approved peptide and protein-based drugs, and hundreds in clinical development [1], [3], [4], [5], [7], [8], [9], [10], [11]. Protein therapeutics comprised 50 % of the top 10 best-selling drugs in 2023 [1]. Peptides including insulin and its analogues, along with GLP-1 receptor agonists (GLP1-RA) account for ∼ 50 % of current peptide therapeutic revenues (total ∼$ 42 billion) [4]. Synthetic and recombinant peptides can exhibit enhanced physiological stability, extended plasma profiles, and higher target affinity than their natural counterparts [5]. Driven by their vast therapeutic potential, approvals of peptide and protein therapeutics have increased over the last six decades, with an average growth rate of 7.7 % for the global peptide therapeutics market [4]. In this review, we focus mainly on peptides.
For prolonged and sustained therapeutic effects, peptide therapeutics are normally injected due to a lack of oral bioavailability [12], [13]. Systemic administration via intramuscular (IM) or subcutaneous (SC) injections are the dominant injection routes due to low physiological barriers to bioavailability and capacity for patient self-administration (for SC). However, for lifelong treatment of chronic diseases such as diabetes, multiple daily- or even weekly SC injections can lead to treatment non-adherence [12], [14], [15], [16]. Beyond the burden of frequent injections, patient-related factors further contribute to non-adherence, including lack of education, self-regulation tendencies, and psychological barriers such as needle phobia and injection pain [17]. Aversion to needles is especially common in paediatric patients, complicating the treatment of children with peptide therapeutics [15]. Approximately 33 % of children with type 1 diabetes have reported a fear of needles, or infusion-site changes due to regular injections [18].
This anxiety can cause non-adherence to treatment, as children with pronounced needle fear tend to have higher HbA1c levels, which could also be due to the reduced blood sugar monitoring frequency, impacting glycaemic control [18]. Some patients with diabetes experience anxiety associated with self-administration of insulin injections, while others may fail to administer the correct insulin dosage [19]. Thus, it is imperative to develop alternative non-injected methods for systemic delivery of peptide therapeutics to improve patient adherence [12], [20]. These include the oral, nasal, transdermal, and buccal routes of administration.
In this context, two particular oral peptide drug formulations converted from parenteral counterparts have recently been commercialized and approved for systemic administration. (Fig. 2). Rybelsus® (semaglutide, Novo Nordisk) was the first oral form of GLP-1 RA approved for the treatment of Type-2 Diabetes (T2D) [21], [22]. Compared to the once weekly SC injection of semaglutide, Ozempic®, the oral formulation of the same molecule is taken daily to achieve long-term glycaemic control [22], [23]. Oral semaglutide is absorbed across the stomach, which is facilitated by the permeation enhancer (PE), sodium N-(8-[2-hydroxybenzoyl] amino) caprylate (SNAC) (Fig. 2A). The sodium salt of this C8 derivative can transiently increase local pH around the tablet, thereby protecting the drug from pepsin-mediated degradation in the stomach while presenting it as a monomer at the stomach wall [21]. An oral formulation of low MW synthetic peptide, octreotide (MW = 1 kDa), Mycapssa®, was approved in 2020 for the treatment of acromegaly and neuroendocrine tumours [20]. The Mycapssa® oily suspension formulation leverages an enteric coating to protect the drug from gastric degradation and a PE, sodium caprylate (C8), presented in an oily suspension to increase intestinal absorption, which transiently opens tight junctions in the intestinal epithelium to facilitate paracellular transport of octreotide into the systemic circulation [24]. Unlike semaglutide formulated as Rybelsus®, octreotide formulated as Mycapssa® is absorbed across the upper small intestine (Fig. 2B), and this route is a favourable alternative to current painful monthly injections of octreotide with low gauge needles, especially for children [10]. Oral formulations of other peptides are in different stages of pre-clinical and clinical development [10].
Fig. 2. Two of the marketed oral peptide formulations for systemic delivery. (A) Rybelsus® was the first oral GLP-1 receptor agonist tablet approved in 2019 for the treatment of Type-2 Diabetes [12]. It has a C18 fatty acid residue conjugated to 31 amino acid long polypeptide chain with approximate MW of 4.1 kDa [11], [23]. Rybelsus® is a co-formulation of semaglutide with N-(8-[2-hydroxybenzoyl] amino) caprylate (SNAC), which facilitates its absorption across the stomach [22], [23]. (B) Mycapssa® was the first oral octreotide capsule approved for treatment of acromegaly in 2020. Octreotide is a synthetic analogue of somatostatin with a MW of 1.02 kDa, which is somewhat more stable due to its cyclic octapeptide structure [20]. Mycapssa® is an oily suspension of octreotide made with the Transient Permeation Enhancer™ (TPE™) technology [20]. Made with BioRender.com.
The oral route poses limitations for peptide absorption. Firstly, despite satisfactory efficacy, the overall bioavailability of Rybelsus® is low at 0.8 %, with a substantial within-subject variability of 137 % [25]. For Mycapssa®, oral bioavailability was estimated to be ∼ 0.7 %.[20]. Poor bioavailability makes oral dosage forms of peptide therapeutics costly despite no requirement for sterility [23]. Secondly, oral absorption of Rybelsus® and Mycapssa® requires abstinence of food for at least 30 min and limited water intake after administration to ensure even that low level of absorption [25]. In addition, Rybelsus® must be administered with no more than 120 ml of water. Thus, there is likely to be adherence issues for patients undergoing chronic daily oral therapy in accepting these dosing restrictions.
Developing formulations for other peptides that protect them from GI degradation while ensuring sufficient permeability remains a significant challenge [26]. Despite advances in multifunctional and stimuli-responsive biomaterials, it is still difficult to significantly enhance oral peptide absorption beyond 1 % with standard permeation-enhancer-based oral dosage forms. Such low bioavailability was only satisfactory for Rybelsus® and Mycapssa® because acceptable efficacy was achieved. For proteins, excluding insulin, the bioavailability levels do not even reach those levels.
Alternatively, receptor-mediated transport mechanisms including the folate receptor and Vitamin B12 pathways have been explored to enhance oral delivery of peptides. Peptides can be conjugated with folate or Vitamin B12 [27], [28], the latter of which uses intrinsic factor-mediated absorption pathways to address poor bioavailability [29]. These approaches demonstrate the potential to significantly improve peptide and protein delivery by providing stability and selective transport, complementing conventional methods; however, to our knowledge, no GI receptor or carrier-targeted system is currently in clinical trials and current preclinical targets for oral peptide nanotechnologies seem to favour the FcRn (neonatal Fc) receptor [30] and the sodium-dependent bile acid transport (ASBT) [31], unlike the SNAC mediated PE [32].
Systemic delivery of peptide therapeutics through other routes is also an alternative to injections and oral administration [33]. Numerous small molecules exhibit absorption across epithelia including the nasal, the buccal cavity, and the vagina [34], [35], [36], [37], [38], [39]. Among these sites, the nasal route has been exploited for the systemic delivery of several peptide therapeutics. For example, desmopressin, a synthetic analogue of vasopressin, is available as a nasal spray for the treatment of nocturia [40]. Additionally, a calcitonin nasal spray is used for the treatment of postmenopausal osteoporosis [41]. A gonadotropin-releasing hormone (GnRH) agonist (nafarelin) nasal spray was developed for the management of endometriosis [42], while a nasal glucagon spray was approved to treat hypoglycaemia in patients with diabetes[43]. These formulations leverage the nasal mucosa’s high vascularization to achieve systemic absorption of peptides. However, several challenges persist in achieving consistent and efficient systemic absorption high MW peptides via the nasal route. Key issues include enzymatic degradation within the nasal cavity, limited absorptive surface area, low acceptability of PE formulations, and variability in mucociliary clearance. Additionally, the MW and hydrophilicity of peptides hinder their permeability across the nasal epithelium [44].
To explore alternative non-invasive delivery routes, the buccal mucosa has emerged as a promising site to administer peptide therapeutics. The buccal mucosa has an extensive network of blood capillaries draining directly into the systemic circulation while avoiding the liver first-pass effect. The oromucosal cavity is lined by sublingual, gingival, palatal, and buccal mucosae. Each of these possess distinct histochemical architecture and biological functions and with differing drug permeability capacities. Several key studies have established benchmarks for buccal peptide delivery [45], [46]. For example, buccal delivery of the oligopeptide, buserelin (1.2 kDa), co-formulated with a bile salt PE, sodium glycodeoxycholate, demonstrated therapeutically relevant systemic levels in pigs [47]. Similarly, peptides such as insulin, oxytocin, vasopressin analogues, protirelin, and octreotide have shown varying degrees of absorption across the buccal mucosa in different animal models, but only with the assistance of suitable PEs [46]. Additionally, chitosan, acting as both a bio-adhesive and a PE, increased the flux of transforming growth factor-beta (TGF-β) across porcine buccal mucosae ex vivo [48].
Several efforts have been made on fabricating mucoadhesive buccal peptide films with chitosan and its derivatives. For example, Cui et al. developed chitosan-ethylenediaminetetraacetic acid- based hydrogel films for delivery of insulin through buccal route. The mucoadhesive hydrogel demonstrated a 17 % pharmacological availability of insulin compared to subcutaneous injection [49]. Similarly, Lancina et al. developed electro spun chitosan nanofibers to significantly enhance the buccal permeability of insulin, demonstrating a 16-fold increase in permeability compared to free insulin [50]. Additionally, Langoth et al. fabricated chitosan–thioglycolic acid (TGA) conjugate-based oromucosal films to enhance the permeability of pituitary adenylate cyclase-activating polypeptide, achieving a 10-fold increase in bioavailability when combined with glutathione [51].
Similarly, Ishida et al. developed a cacao butter-based buccal formulation for insulin delivery. This was co-formulated with sodium glycocholate as a PE and a hydroxypropyl cellulose (HPC)–Carbopol® mixture in 1:2 ratio to enhance the adhesion, dissolution, and fracture resistance [52]. An in vivo study in dogs reported 0.5 % bioavailability of insulin compared with intramuscular route, demonstrating low systemic absorption. Schurr et al. evaluated a self-adhesive buccal patch containing Thyrotropin-Releasing Hormone (TRH) among healthy subject [53]. Buccal TRH patch resulted in a gradual TSH increase with maximal TSH plasma levels achieved after 120 min. Veillard et al. developed a bio adhesive buccal patch for a lauryl derivative of a tripeptide, consisting of a polycarbophil-based mucoadhesive membrane, a rate-limiting membrane, and an impermeable backing layer [54]. Furthermore, Hoogstraate et al. investigated the buccal delivery of buserelin using a Hilltop™ chamber supported with an adhesive backing [55]. The in vivo study showed that sodium glycodeoxycholate (GDC) at 10 mM concentration, increased buserelin bioavailability from 1.0 % to 5.3 %, demonstrating the permeation enhancement effect of bile salts.
Several other studies have explored buccal patches with varied compositions for peptide delivery, with differences in permeation enhancement and bioavailability. Li et al. developed a Carbopol 974P-silicone polymer buccal patch for buccal delivery of oxytocin, demonstrating a steady-state flux of oligopeptide in rabbits with 0.1 % bioavailability [56]. Lee and Chien developed a bilayer mucoadhesive patch for LHRH. Its design consist of a fast-release layer (PVP K-30) and a sustained-release layer (Carbopol 934 and PVP K-90), achieving controlled release [57]. Similarly, Nakane et al. developed a bilayer transmucosal therapeutic system for LHRH delivery, which was designed for controlled and enhanced absorption through the oromucosal route [58]. The fast-release layer contained PVP K-30, with cetylpyridinium chloride, and bile salts (sodium taurodeoxycholate, sodium deoxycholate, or sodium cholate) as PEs, facilitating rapid drug release and improved absorption. Whereas, the sustained-release layer was composed of Carbopol 934P and PVP K-90, which provided prolonged drug release. In vivo studies in Beagle dogs demonstrated that incorporating 5 % bile salt in the fast-release layer significantly enhanced bioavailability (up to 1.6 %), achieving higher plasma levels and sustained systemic absorption. The study highlighted that bile salts effectively increased transmucosal permeation, while the bilayer design ensured rapid onset followed by sustained release, making it a promising system for buccal peptide drug delivery.
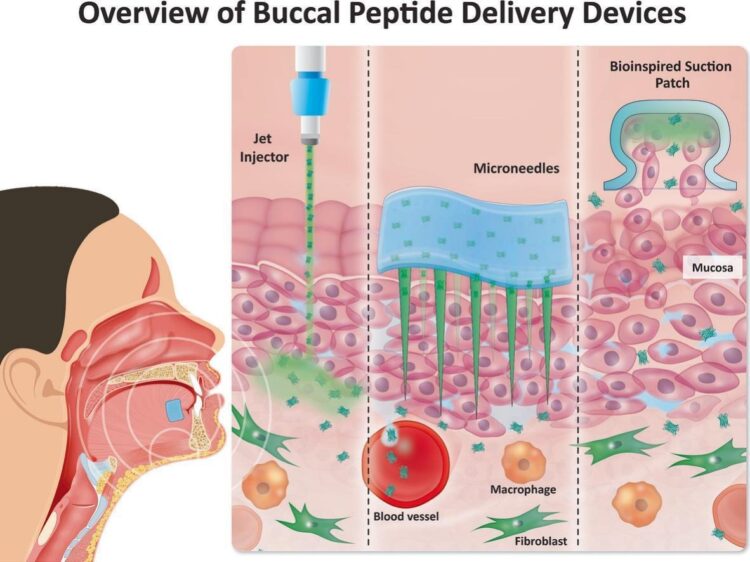
For oromucosal delivery of peptide therapeutics, similar to the stomach and upper small intestine, the human non-keratinized buccal epithelium restricts diffusion. However, attempts to commercialize mucoadhesive buccal films for peptide and protein therapeutics have not yet had success. One of the main reasons is insufficient permeability of peptide therapeutics across oromucosal tissue in an acceptable time frame. This review is focused on the development of buccal patches/films for systemic delivery of therapeutic peptides using physical device strategies to overcome the physiological barrier. Furthermore, we highlight future design considerations, along with commercialization and regulatory challenges that need to be addressed in translating buccal peptide delivery devices for clinical use.
Download the full article as PDF here Devices to overcome the buccal mucosal barrier to administer therapeutic peptides
or read it here
Table 1. Commercialized buccal/sublingual films for small molecules (selected).
| Product | Manufacturer | Polymers | Molecule | Indication |
|---|---|---|---|---|
| Belbuca® | BioDelivery Sciences International Ltd | CMC, Polycarbophil, HPC, HEC [78] | Buprenorphine | Breakthrough pain |
| Bunavail® | BioDelivery Sciences International Ltd | CMC, HEC, HPC, Polycarbophil [68] | Buprenorphine/ Naloxone | Opioid dependence |
| Libervant™ | Aquestive® Therapeutics Inc | Hypromellose, PEO, Xanthum Gum, PVP [79] | Diazepam | Breakthrough seizures |
| Suboxone® | Indivior plc | PEO, HPMC [69] | Buprenorphine/ Naloxone | Opioid dependence |
| Onsolis® | BioDelivery Sciences International Ltd | CMC, HEC, HPC, Polycarbophil [67] | Fentanyl | Breakthrough pain |
| Wafesil™ | iX Biopharma Ltd | Cellulose, Carmellose [80] | Sildenafil citrate | Erectile dysfunction |
| Zuplenz® | Galena Biopharma | HPMC, PEO [71] | Ondansetron | Nausea and vomiting |
Sahil Malhotra, Thomas Lijnse, Eoin O’ Cearbhaill, David J. Brayden, Devices to overcome the buccal mucosal barrier to administer therapeutic peptides, Advanced Drug Delivery Reviews, Volume 220, 2025, 115572, ISSN 0169-409X, https://doi.org/10.1016/j.addr.2025.115572.









































 All4Nutra
All4Nutra









