CBD Oil Tablets Chewable – Direct Compression with PROSOLV 730

CBD Oil Tablets Chewable – Direct Compression with PROSOLV 730
Aim of the Study
Due to the physico-chemical properties of cannabidiol (CBD), the vast majority of the marketed products containing CBD are oils. To provide greater convenience in dosing, the goal of this experiment was to make directly compressible 25 mg CBD oil tablets as well as 25 mg CBD oil chewable tablets with minimal effort.

CBD Oil
Cannabidiol (CBD) is a non-psychotropic compound found in the cannabis plant. Its anti-inflammatory, anti-anxiety, anti spasmodic, and anti-nausea properties make CBD an increasingly popular natural remedy used for relieving uncomfortable symptoms of many common diseases. Further pharmacological effects are being researched, making CBD an appealing option for new therapies.[1;2]
The CBD oil used in this study was a thick to black tar dark green like substance (Figure 1) which would have been difficult – if not impossible – to work with, using conventional tableting excipients.

Formulations
Two different solid dose formulations were made, combining the CBD oil with the high functionality adsorbent binder PROSOLV ® 730 and other JRS excipients depending on the formulation as described in the table below. All formulations produced 25 mg CBD oil tablets.
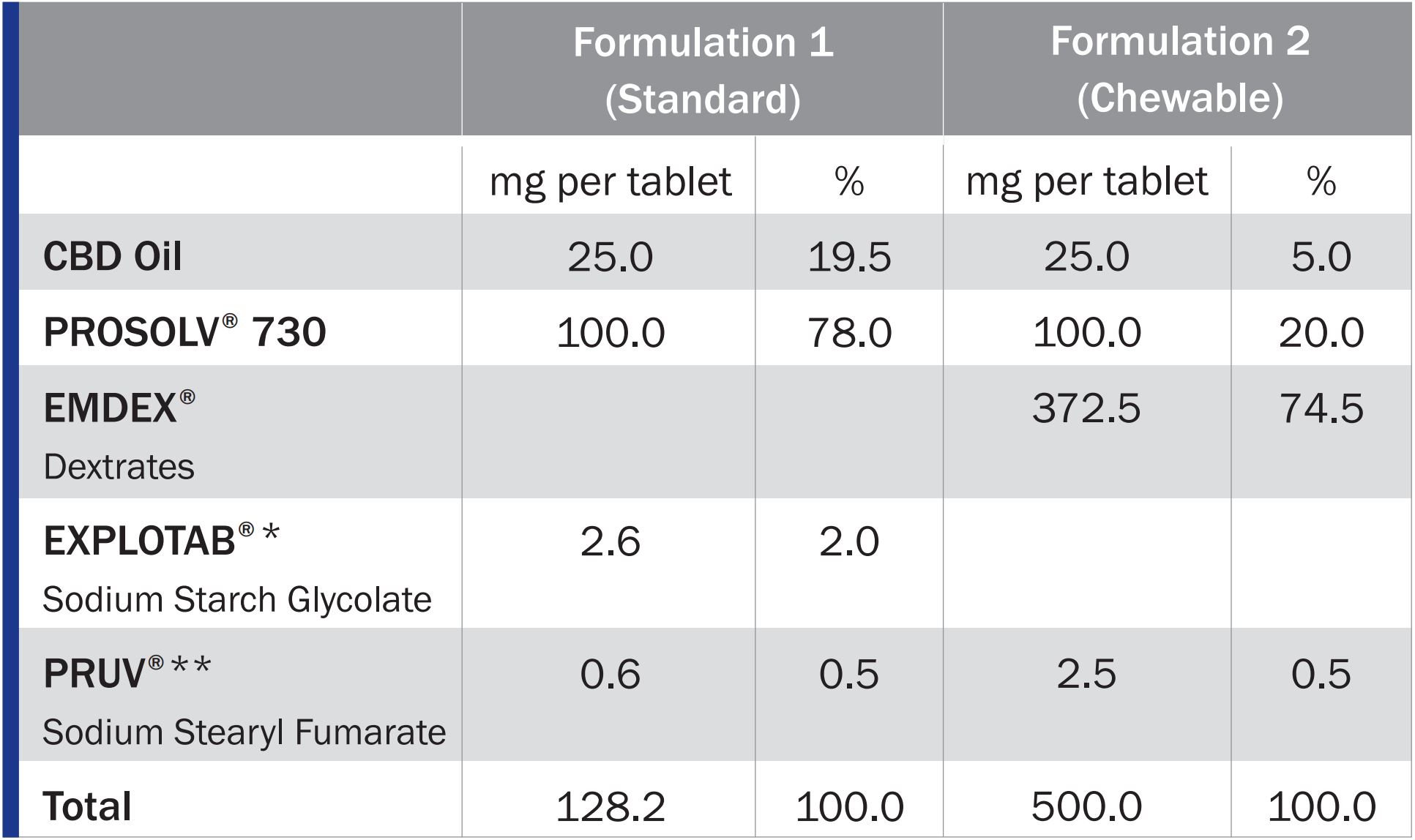
The CBD oil was obtained from HempMeds.
Excipients used
PROSOLV® 730 is a high functionality excipient that was specifically designed for the direct compression or encapsulation of oily active ingredients or lipophilic actives dissolved in oil.
Combining PROSOLV 730 ® with CBD oil and additional excipients, such as EXPLOTAB® sodium starch glycolate as disintegrant, and PRUV® sodium stearyl fumarate or LUBRI-PREZ magnesium stearate as lubricant conveniently enables direct compression of standard, immediate-release tablets.
Chewable tablets can be produced by direct compression in an equally straightforward way by combining CBD-loaded PROSOLV® 730 with EMDEX® dextrates as a naturally sweet and highly soluble binder. Due to its neutral taste, PRUV® sodium stearyl fumarate is the preferred lubricant for chewable formulations. If required due to local food regulations, LUBRI-PREZ magnesium stearate can is a suitable alternative.
Procedure
Blending
The CBD oil was added to PROSOLV® 730 via high shear mixing for 10 minutes. The oil-loaded PROSOLV® 730 was then transferred to a low shear mixing vessel and blended for 10 minutes with the remaining excipients, except the lubricant. PRUV® sodium stearyl fumarate was sieved through a 20 mesh screen and subsequently added to the low shear mixer and blended for an additional 5 minutes. This blend was then immediately used for direct compression tableting.
The CBD oil can first be heated to 50°C in a water bath for 10 minutes to reduce the viscosity of the material if necessary. The formulations presented here were made with and without heating the CBD oil with no difference in results.
Equipment
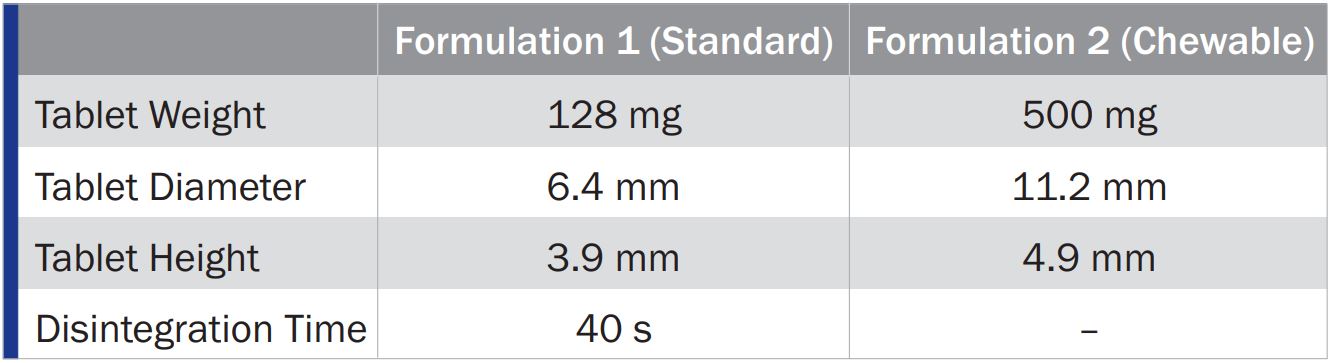
Tablet Characteristics at 15 kN Compaction Force
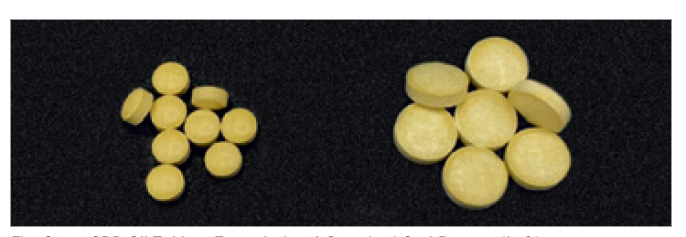
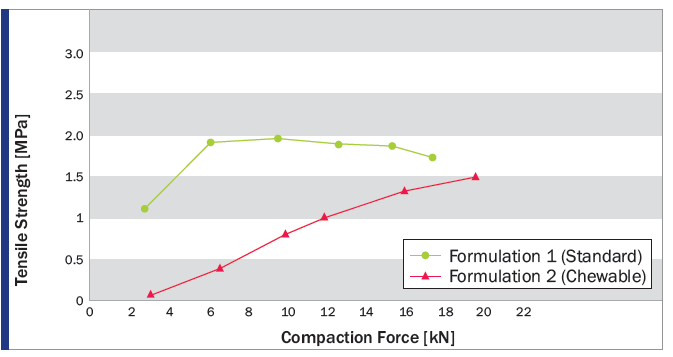
made with PROSOLV® 730
Conclusion
PROSOLV® 730 was found to be perfectly suited for the production of CBD oil tablets by direct compression. Both dosage forms showed good overall tabletability. The standard orally ingestible tablets (Formulation 1) had the highest tensile strength, lowest ejection forces, and fast disintegration times. The chewable tablets (Formulation 2) exhibited the physical properties, desirable for this type of tablet.
PROSOLV® 730 enables simple, direct compression formulation of CBD Oil, thus offering the possibility of producing different solid dosage forms.
See and download the White Paper “CBD Oil Tablets Chewable” here
(click the picture to download the White Paper)
Source: JRS Pharma White Paper “CBD Oil Tablets Chewable”
References
[1] P. Nagarkatti et. al., Future Med Chem. 2009 Oct; 1(7): 1333–1349.
[2] A.W. Zuardi et. al., Braz J Med Biol Res 2006 Apr; 39 (4).
Do you need more information or a sample of JRS Pharma excipients?









































 All4Nutra
All4Nutra








