Formulating Cannabinoids

Gattefosse CBD
Cannabinoids are challenging APIs to formulate:
- Being highly lipophilic compounds with low water solubility, cannabinoids need to be solubilized for subsequent absorption in the gastro-intestinal tract
- With high first-pass metabolism, significant food effect and high potency, bioavailability of cannabinoids is highly dependent on the formulation
- Available as powder, liquid, resin, their physical state may require specific preparation to be incorporated in the dosage form
- With high sensitivity to light, temperature and oxidation, special care has to be taken during handling, processing and storage
Gattefossé has excipients to formulate high quality tinctures, drops, sprays, tablets, creams, gels, suppositories
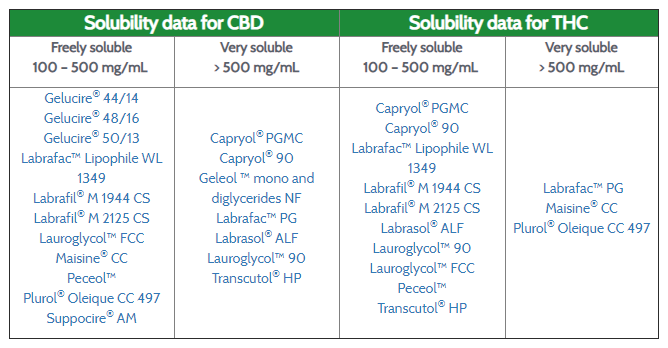
High solubilizing capacity of lipid excipients
All our excipients show exceptional solubilizing capacity for cannabinoids and ensure the solubilization of the therapeutic dose.
 Enhancing oral bioavailability of poorly-water soluble drugs with lipid-based formulations
Enhancing oral bioavailability of poorly-water soluble drugs with lipid-based formulations
Lipid-based formulations solubilize and maintain the drug in solubilized state throughout the gastro-intestinal tract, leading to normalized drug absorption and increased bioavailability. Reduction of food effect, increase in lymphatic transport or increase in intestinal permeation are recorded depending on the components of the formulation.
Different types of formulations can be prepared from simple oily formulation to self-emulsifying drug delivery systems depending on the final dosage forms (tinctures, drops, capsules…).
Find out more information on our range of excipients for bioavailability enhancement and lipid-based formulations development. Link to Gattefossé “Formulating cannanbinoids” . Other information – click here or the picture.









































 All4Nutra
All4Nutra








