An audit of pharmaceutical continuous manufacturing regulatory submissions and outcomes in the US
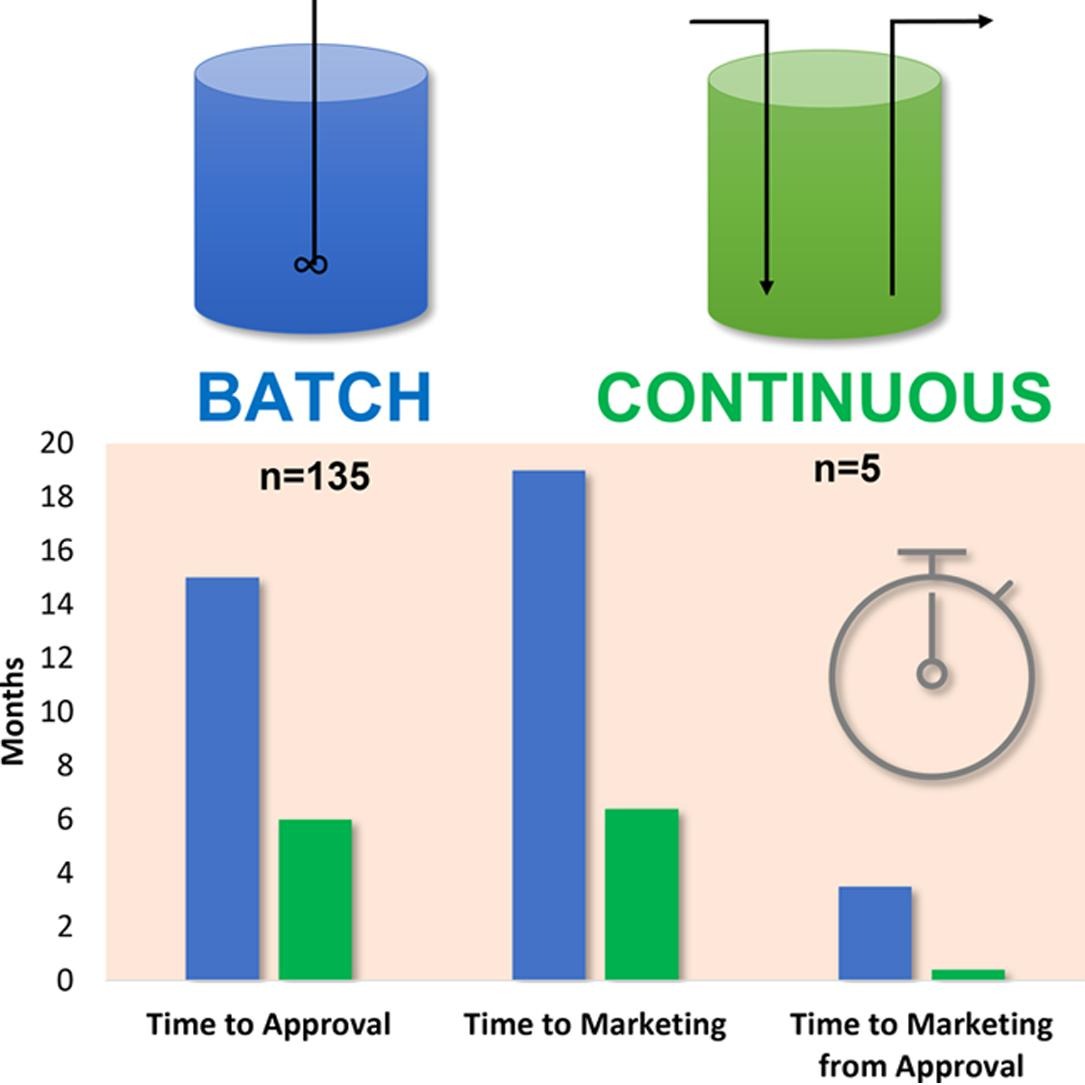
Continuous manufacturing (CM) sends materials directly and continuously to the next step of a process, eliminating hold times and reducing processing times. The potential benefits of CM include improved product quality, reduced waste, lower costs, and increased manufacturing flexibility and agility. Some pharmaceutical manufacturers have been hesitant to adopt CM owing to perceived regulatory risks such as increased time to regulatory approval and market entry, more difficulty submitting postapproval changes, and higher inspectional scrutiny. An FDA self-audit of regulatory submissions in the U.S. examined the outcomes, at approval and during the product lifecycle, of continuous manufacturing applications as compared to traditional batch applications. There were no substantial regulatory barriers identified for CM applications related to manufacturing process changes or pre-approval inspections. CM applicants had relatively shorter times to approval and market as compared to similar batch applications, based on the mean or median times to approval (8 or 3 months faster) and marketing (12 or 4 months faster) from submission, translating to an estimated $171–537 M in early revenue benefit.
Download the full research paper as PDF: An audit of pharmaceutical continuous manufacturing regulatory submissions and outcomes in the US
Adam C. Fisher, William Liu, Andreas Schick, Mahesh Ramanadham, Sharmista Chatterjee, Raphael Brykman, Sau L. Lee, Steven Kozlowski, Ashley B. Boam, Stelios C. Tsinontides, Michael Kopcha,
An audit of pharmaceutical continuous manufacturing regulatory submissions and outcomes in the US,
International Journal of Pharmaceutics, Volume 622, 2022, 121778, ISSN 0378-5173,
https://doi.org/10.1016/j.ijpharm.2022.121778.

