β-cyclodextrin-based multifunctional carriers for colon-targeted drug delivery: advances in hydrogel systems and mucoadhesive strategies

Schematic overview of therapeutic strategies using β-cyclodextrin for colon-targeted drug delivery.
Oral drug delivery to the colon remains a formidable challenge due to the harsh conditions of the upper gastrointestinal tract, including variable pH, enzymatic degradation, and limited mucosal permeability. β-Cyclodextrin (β-CD), a cyclic oligosaccharide with a hydrophobic cavity and hydrophilic exterior, offers a promising approach to overcome these barriers by forming stable inclusion complexes that protect sensitive drugs and modulate their release profile.
This review explores the use of native and chemically modified β-CDs as functional carriers for colonic drug delivery, with a focus on hydrogel-based systems, mucoadhesive derivatives, and microbiota-responsive formulations. A critical review of over two decades of literature was conducted to evaluate the design, performance, and pharmacokinetic benefits of β-CD-loaded systems for intestine-specific therapeutic delivery. Evidence shows that β-CD hydrogels loaded with anti-inflammatory drugs, chemotherapeutics, and poorly soluble molecules achieve pH-sensitive, sustained drug release with improved mucosal adhesion and enhanced bioavailability. Thiolated β-CDs and hydroxypropyl derivatives improve the intestinal residence time and facilitate permeation across the mucus barrier.
In addition, S-protected thiolated β-CDs and NAC/arginine-modified β-CDs exhibit superior mucoadhesion and epithelial absorption, offering prolonged gastrointestinal retention. Formulations incorporating microbiota-triggered release mechanisms exhibit targeted activation in the colon due to fermentation/degradation of β-CDs by colonic Bacteroides, thereby reducing the systemic side effects. The manuscript also critically examines the application of β-cyclodextrin and its derivatives in advancing colon-targeted drug delivery through pH-responsive hydrogels synthesized via structural modifications, such as thiolation and hydroxypropylation that significantly enhance mucoadhesion, residence time, and drug permeation across intestinal barriers.
The integration of β-CD with biopolymers and smart release triggers enables a controlled, site-specific therapeutic release with reduced systemic toxicity and improved pharmacokinetics. Overall, β-CD-based carriers enable multifunctional, site-specific delivery to the colon through a combination of physicochemical stabilization, mucoadhesion, and enzymatic responsiveness. Their application spans both small molecules and biologics, positioning β-CD as a foundational scaffold in the development of next-generation oral therapeutics for gastrointestinal disorders.
See the full article as PDF: β-cyclodextrin-based multifunctional carriers for colon-targeted drug delivery
Introduction
Intestine targeted drug delivery is usually impaired by various physiological barriers which either result in a premature drug release or adversely impact the drug absorption thereby impeding the overall drug pharmacokinetics. Orally administered drugs counter several physiological barriers, including pH variations (acidic in stomach, neutral in small intestine, and slightly acidic to neutral in colon) which may result in off target, premature burst release of the cargo drug (Fig. 1), exposing the patient to a local or a temporal drug overdose (Homayun et al. 2019). Further adverse effects of such events include ulcerogenicity due to an increased local concentration of the drug. Similarly, the thickness and charge of GI mucosa present on the inner epithelia of intestine also present a sizeable challenge in form of a physiological barrier (Fig. 1) which hinders the accomplishment of an optimal drug pharmacokinetics due to a limited drug absorption across the colon (Peterson and Artis 2014). In addition to this, a higher water absorption capacity of colon, and a considerably high viscosity of the colonic contents limit the availability of most of the drugs to the absorptive membrane of the colon (Philip and Philip 2010).
Furthermore, the enzymatic degradation of drugs and their delivery vehicles by the lipases, amylases, and proteases of upper GI tract present another limitation for colon targeted drug delivery as these factors cause inactivation of the drug by lysing/degrading the enzyme-responsive bonds in the drug molecule itself or its carrier system (Fatima et al. 2023). Variability in the transit time of administered drug from the upper GI tract to the colon in diseased states such as ulcerative colitis and Crohn’s disease, circadian rhythm, dietary status, and age is another important factor which determines the intestinal drug absorption (Vinarov et al. 2021). Hence, the need for the development of innovative colon targeted delivery systems which effectively counter these intricacies is quite compelling for improved patient compliance and for ensuring an ideal therapeutic effect of the cargo pharmaceutical.
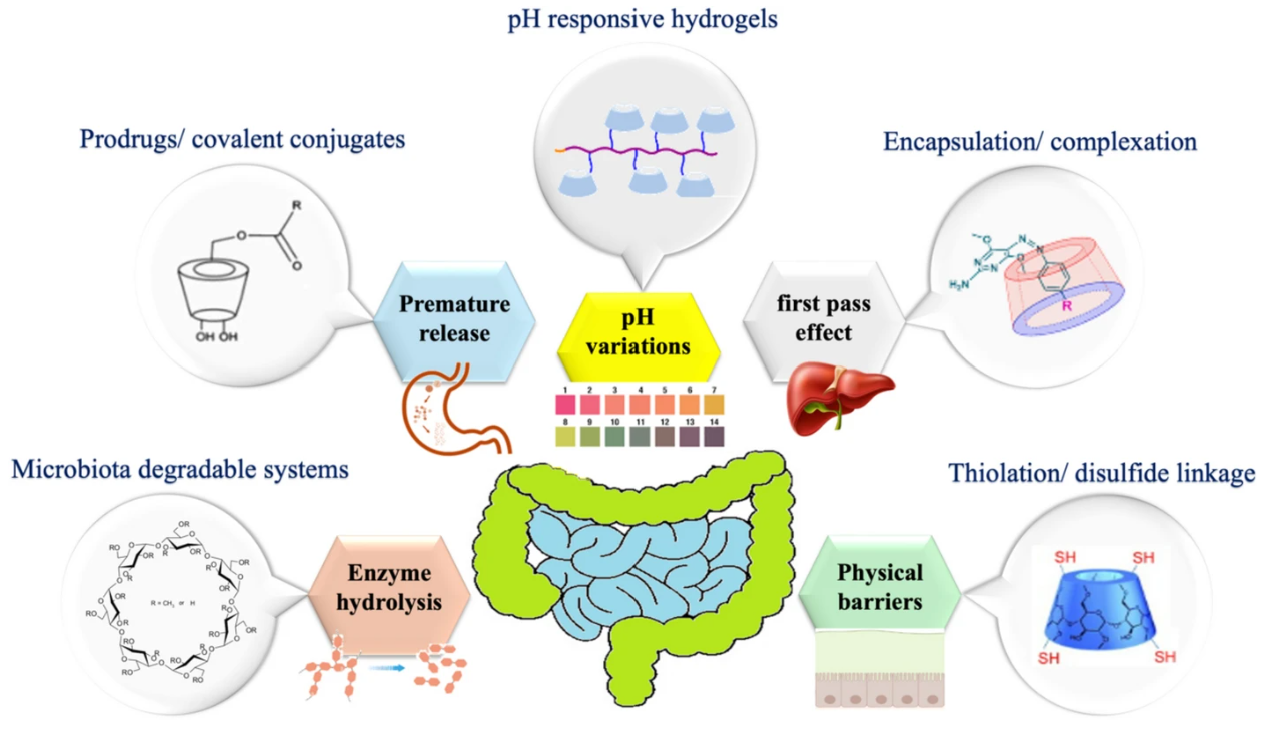
β-cyclodextrin (β-CD) consists of a circular arrangement of seven glucose molecules which are connected via α-1,4 linkages to generate a toroid shape with primary and secondary hydroxyl groups at the opposite rims of the truncated structure (Sandilya et al. 2020). Neutron diffraction studies have shown that the presence of flip-flop hydrogen bonding between the secondary hydroxyl group in β-CD rim that imparts structural rigidity and reduces the hydrophilicity of the macrocycle (Langan et al. 1999). Among the native CDs, β-CD having an appropriate cavity size of 6.0–6.5Å (small cavity size of 4.7–5.3Å for α-CD, and large cavity size of 7.5–8.3Å for γ-CD), a better complexation ability with a variety of ligands (γ-CD possesses the poorest), and its cost effectiveness (as compared to the expensive α-CD) presents an ideal candidature for the encapsulation and delivery of a wide range of cargo drug molecules.
Stability towards non-enzymatic hydrolysis, and resistance towards enzymatic degradation by amylases further prove useful for the generation of physiologically stable inclusion complexes of β-CD for intestine targeted drug delivery applications (Przybyla et al. 2020). Apparently, these inclusion complexes mostly pass the upper gastrointestinal tract to reach the colon, where the microbiota initiates the fermentation of β-CD to achieve a rapid release of the cargo drug molecules which is implicated for the treatment of diseases, such as colorectal adenocarcinoma, Crohn’s disease, ulcerative colitis, and other infections.
Finally, the derivatization of β-CD with mucoadhesive/-mucus penetrating polymers, such as chitosan and alginate, and by the immobilization of thiol/-amine containing ligands on β-CD backbone has been reported to overcome the mucosal barrier for a better absorption of the payload pharmaceuticals (Kamphuis et al. 2017). In the present review article, we discuss about the colon targeted drug delivery with β-CD-based carrier systems, including hydrogels, mucoadhesive derivatives, and microbiota (colon) digestible formulations. Figure 1 presents a schematic summary of the mechanistic pathways through which β-cyclodextrin-based systems achieve site-specific intestinal drug delivery.
Continue reading here: β-cyclodextrin-based multifunctional carriers for colon-targeted drug delivery
Sharma, M., Prasher, P., Fatima, R. et al. β-cyclodextrin-based multifunctional carriers for colon-targeted drug delivery: advances in hydrogel systems and mucoadhesive strategies. Cancer Nano 16, 36 (2025). https://doi.org/10.1186/s12645-025-00339-w









































 All4Nutra
All4Nutra







