Elucidation of the effect of added fines on the performance of dry powder inhalation formulations
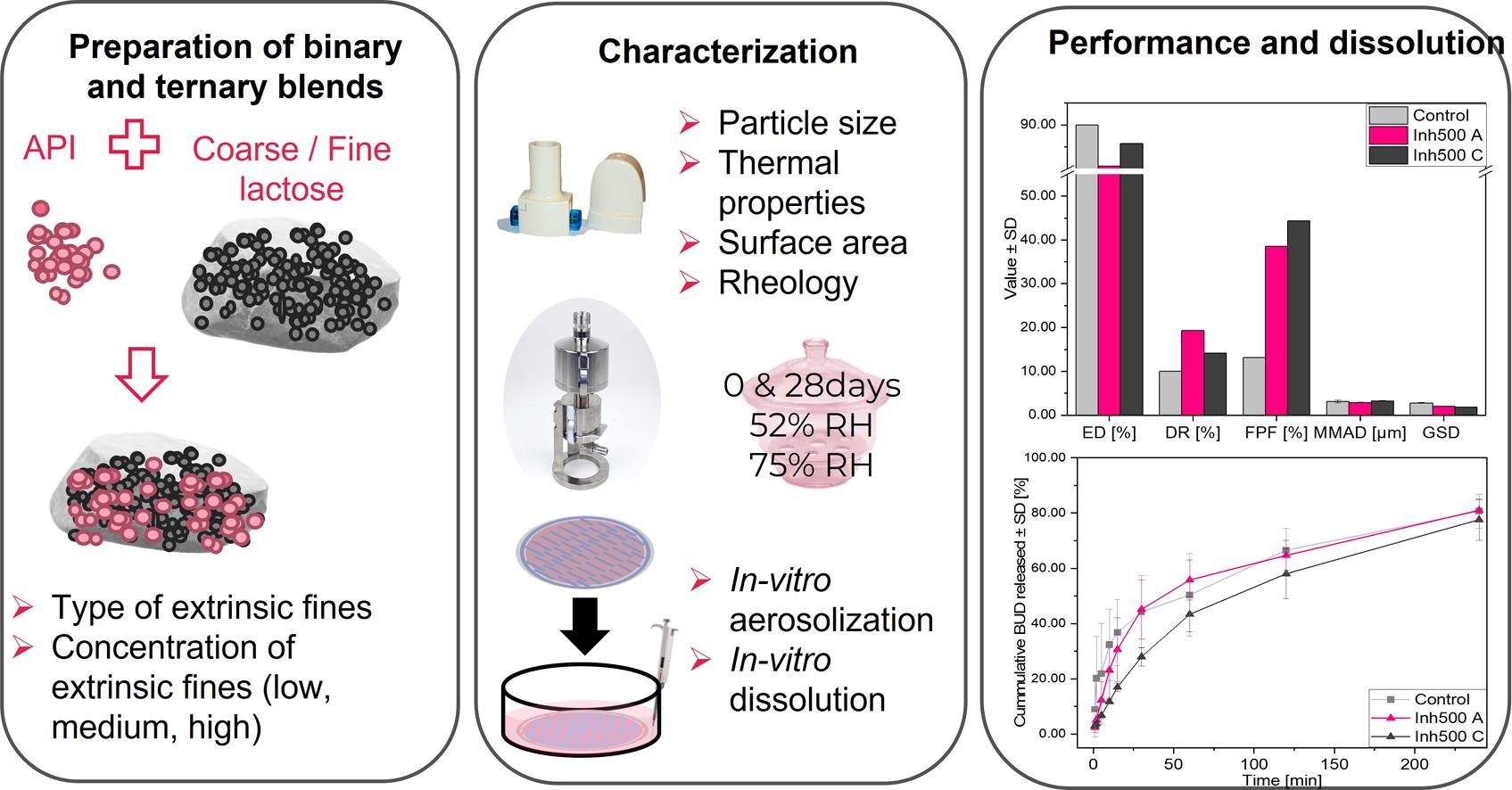
Dry powder inhalers (DPIs) are regularly used to treat respiratory diseases. Adding extrinsic fine excipient particles to the blend of active pharmaceutical ingredient (API) and carrier is an established strategy to improve aerosolization efficiency during pulmonary drug delivery. Different amounts and grades of lactose fines may, however, compromise the flowability and downstream processing of the material. Further, given the particle size of the inhaled fine particles (<5.5 µm), also deposition of lactose fines to different lung regions following inhalation cannot be excluded. This study aimed to investigate the impact of commercially available extrinsic lactose fine materials produced using different milling parameters, on physicochemical properties and aerosolization performance of ternary blends, as a factor of time and storage conditions.
Highlights
The addition of extrinsic fines:
• Improved aerosolization efficiency of DPI formulations drastically.
• Increased cohesivity of powder formulations.
• Impacted (retarded) dissolution of Budesonide from ternary blends intended for inhalations.
Further, for the first time, it was attempted to elucidate the effect that the amount of present fines has on the dissolution of the model API from the ternary blends exposed to different storage conditions. We showed that rheological behavior was impacted when a higher amount of fines was present, and this effect was further enhanced by storage at high relative humidity. The aerosolization efficiency was vastly improved with increasing content of fines. Still, initial data indicated that the dissolution of the poorly soluble API was retarded when more fines were present in blends.
Lactose excipient mentioned in the study: Inhalac 500
Milica Stankovic-Brandl, Snezana Radivojev, Pia Sailer, Franz-Karl Penz, Amrit Paudel, Elucidation of the effect of added fines on the performance of dry powder inhalation formulations, International Journal of Pharmaceutics,
Volume 629, 2022, 122359, ISSN 0378-5173, https://doi.org/10.1016/j.ijpharm.2022.122359.
See the overview video of the Meggle Inhalac product range DPI here:

