Streamline Your R&D and Production
Introduction
The health science industry faces a wide variety of challenges including increased pressure to:
- Reduce costs
- Accelerate time to market
- Improve product performance
- Cope with high demands and tight capacities
- Utilize modern technology
These challenges drive health science companies to find new solutions for improving time – and cost–effciency in oral dosage form development and production. Formulation scientists developing direct compression formulations generally require several conventional excipients – often at high levels – to obtain good material flow, compaction, blending properties, content uniformity, carrying capacity, stability, lubricity, and disintegration. Finding the proper excipient combinations and concentrations to achieve adequate solid dosage formulas is a timeconsuming, and often expensive process, that varies with active pharmaceutical ingredient (API) characteristics.
PROSOLV® RX 90 was developed to provide an innovative solution to meet these challenges.
Beyond its benefits in conventional batch production, PROSOLV® RX 90 presents a set of properties that make it well-suited for continuous manufacturing (see page 9). Because it acts multifunctionally as a filler/binder, flow aid, disintegrant, and lubricant at the same time, production requires only two feeders. This not only leads to space saving, but also helps to simplify control of the production process.
The unique surface structure and good flowability of PROSOLV® RX 90 enable fast and segregation-free blending with APIs.
PROSOLV® technology
Compounding with the PROSOLV® Technology leads to a homogeneous distribution of the individual components throughout the particle and on its surface.
The synergistic effects achieved by this technology include better compactability, flowability, and content uniformity, due to a significantly increased surface area.
Furthermore, its all-in-one structure provides outstanding robustness in terms of blending times:

What is PROSOLV® RX 90?
PROSOLV® RX 90 was developed using JRS PHARMA‘s proven PROSOLV® Technology and was the first lubricated high functionality excipient (HFE) on the market.
PROSOLV® RX 90 is an all-in-one, ready-touse excipient composite. It effectively combines all functionalities required for tableting in a single excipient composite.
PROSOLV® RX 90 is more than a simple physical blend. It is a homogeneous, lubricated high functionality excipient composite. Each component of the composite maintains its chemical identity while synergistically providing increased functional performance.
6 Elements of Success
Powder flow, blend homogeneity and good compactability are prerequisites for successful tableting.
Mechanical robustness of tablets enables dust-free coating and packaging.
Fast disintegration is essential for in-vivo performance of the tablet. PROSOLV® RX 90 combines these 6 elements of success in one excipient:
1. Excellent powder flow
Due to its favorable particle morphology, PROSOLV® RX 90 shows significantly better powder flow properties than the corresponding physical mixture of its individual ingredients.
PROSOLV® RX 90 is therefore perfectly suited for direct compression processing, even on high speed tablet presses.
2. Superb blend homogeneity
The composite nature of PROSOLV® RX 90 prevents segregation of the individual excipient components.
Moreover, the porous surface structure promotes adhesion of low-dose, micronized active ingredients, thus improving the content uniformity in the powder blend and the finished product.
3. Outstanding compactability performance
The excellent compaction and lubrication of PROSOLV® RX 90 result in less capping and breakage, increasing yields and improving profitability. With PROSOLV® RX 90, mechanically robust tablets can be produced at a low compaction force. This leads to prolonged equipment and tooling life.
Due to a smaller excipient amount needed and the possibility of high active ingredient loads, smaller tablet sizes and lower tablet weight can be reached, which results in high patient compliance.
4. Ultimate robustness, improved tablet handling
PROSOLV® RX 90 tablets are mechanically robust and have a low friability, even in the case of demanding tablet shapes. Tablet robustness enables all packaging and coating options. PROSOLV® RX 90 tablet production generates less dust. Tableting with PROSOLV® RX 90 can reduce cleaning and change – overtimes (see pages 7 and 8 of the Flyer below).
5. Rapid disintegration
The built-in superdisintegrant enables rapid disintegration with minimum variation from tablet to tablet.
6. Ideal for film coating
Due to its unique surface structure, the PROSOLV® RX 90 line is ideally suited for film-coated tablets. It enables crisp logo definition and excellent film adhesion to the core.
PROSOLV® RX 90 Composition

Benefits of PROSOLV RX 90
Traditional tablet production methods include dry granulation, wet granulation, and direct compression. Granulation processes are typically complex, requiring a great deal of equipment, human resources, and time. The lengthy, multi-stage process leaves room for error, and thus, batch losses. Direct compression is a shorter, and often preferred, production process. It requires fewer resources than wet granulation and leaves less room for error and batch loss (Figure 3).
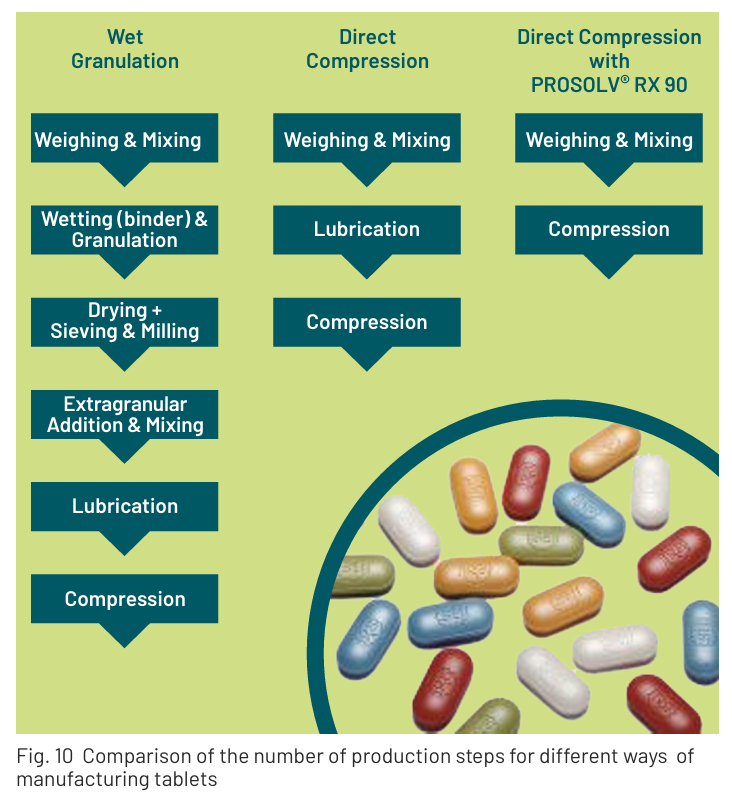
The PROSOLV® RX 90 line includes all the necessary excipients for direct compression in an all-in-one composite. Simply add the APIs, blend and compress into tablets. This easy method of tableting allows for shorter development times and lower R&D costs, leading to faster time to market. The outstanding compaction properties enable smaller tablet sizes, faster production and may boost profitability due to prolonged tooling life.
With PROSOLV® RX 90, buy, analyse and store only one excipient instead of five, thus reducing storage and quality control costs.
Conclusion:
- GMO-Free
- Allergen-Free
- BSE/TSE-Free
- Complies with general chapters for residual solvents › (USP <467>, Ph. Eur. 5.4)
- Re-evaluation date: 3 years
- US DMF available
- Full regulatory package available
- Regulatory information available for registration in › all major markets.
See case studies in the full technical brochure on “PROSOLV® RX 90” here
(click the picture to download the brochure)
Source: JRS Pharma technical brochure “PROSOLV® RX 90”
Do you need more information or a sample of JRS Pharma excipients?









































 All4Nutra
All4Nutra














