Days or Centuries? – Predicting the Shelf-Life of ASDS

Predicting the Shelf-Life of ASDS
PURPOSE
Amorphous solid dispersions (ASDs) are a state-of-the-art enabling formulation technique for poorly water-soluble drugs. Drug recrystallization in the polymer matrix highly unwanted: marks end of shelf life of such a formulation.
- Overcoming the poorly understood drug recrystallization risk during storage and random stability screenings
- Understanding the impact of different storage conditions (temperature, RH) and packaging alternatives
- Predicting shelf life at any drug load/ temperature/humidity condition ahead of the stability test with novel in-silico tool and minimal data set
OBJECTIVE
Develop and validate predictive model for shelf life considering thermodynamic factors (water absorption, fundamental crystallization driving forces), kinetic factors (glass transition, diffusivity in the ASD), drug-specific crystallization mechanism (nucleation, crystal growth, glass-forming ability), thermal history (spray drying, melt extrusion, glass aging) and their mutual impact.
METHODS
RESULTS
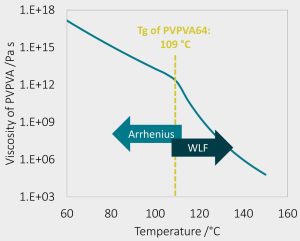
Viscosity model for the ASD matrix
Understanding kinetic factors of ASD shelf life
- Diffusivity within ASD matrix governed by polymer viscosity
- PVPVA64 viscosity above Tg obtained from literature
- Time/ Temperature Superposition principle: Only Tg of ASD required to determine ASD viscosity
- Analogous principle applied to wet Tg/viscosity at elevated RH
Thermodynamic model PC-SAFT
Understanding thermodynamic factors of ASD shelf life
- Equilibrium felodipine solubility in PVPVA64, felodipine crystallization driving force, moisture sorption and the mutual impact predicted (PC-SAFT)
- Equilibrium solubility dramatically reduced at room temperature
Stability prediction – days or centuries?
Felodipine/PVPVA64 ASD stability at moderate storage conditions predicted as function of felodipine load in ASD:
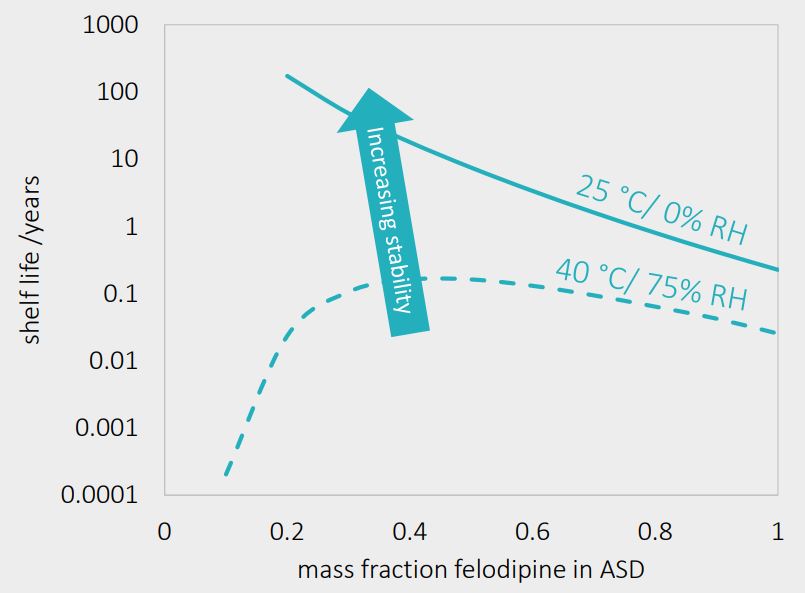
The predicted shelf life ranges from few days at 40C/ 75% RH (max. 0.16 years at 40w% felodipine load) to centuries at 25C/0% RH (172 years at 20wt% felodipine load and 25C /0% RH)
Crystallization model
Understanding nucleation and crystal growth regime
- Measuring crystallization kinetics in pure felodipine or high felodipine loaded ASDs at high T/RH conditions
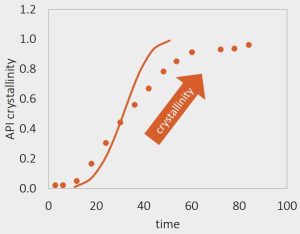
- Crystallization kinetics measured in pure API/ High API/ High T/R
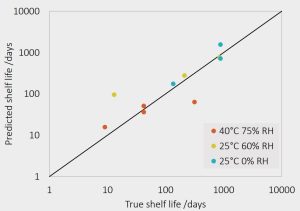
Validation of the in-silico tool
- Long-term storage of spray-dried ASDs with felodipine content between 10w% and 90 w% at the storage conditions 25C / 0% RH, 25C/ 60% RH and 40C/ 75% RH for three years
- Crystallization onset time determined via repeated X-ray diffraction analysis
CONCLUSIONS
- Novel in-silico model has been successfully applied for predicting the shelf life of metastable ASDs
- Shelf-life predictions validated by three-years stability tests at three storage conditions
- Few key experiments instead of random screenings
- Stability data collected at so-called ‘accelerated’, harsh storage conditions within short time do not at all reflect the shelf-life at moderate storage conditions as thermodynamic and kinetic aspects differ completely
➢ The developed model can extrapolate ‘harsh data’ better as it accounts for the changes in thermodynamic and kinetic aspect
For more information download the full poster as PDF: “Days or Centuries? – Predicting the Shelf-Life of ASDs“:
(click the picture below)
Source: Poster “Days or Centruries? – Predicting the Shelf-Life of ASDS” by aaps PharmSci360, Christian Luebbert, Gabriele Sadowski, amofor GmbH









































 All4Nutra
All4Nutra








