Topical delivery of nanoemulsions for skin cancer treatment
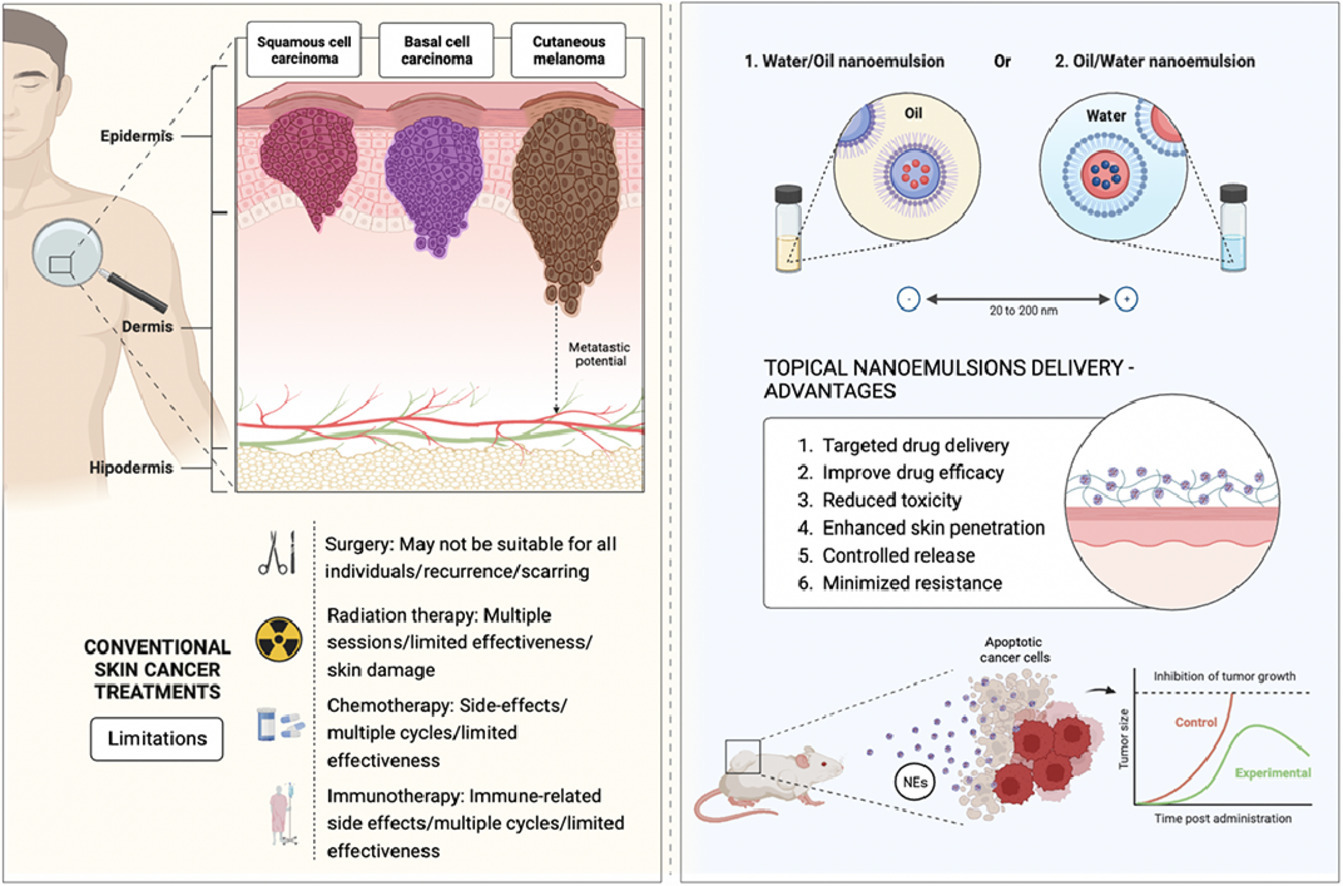
Skin cancer chemotherapeutics often lead to the development of severe cytotoxicity, compelling the development of novel delivery systems to not only enhance therapeutic efficacy but also minimize side effects and improve patient compliance. In recent years, topical nanoemulsions have emerged as powerful tools in the field of skin cancer therapeutic management. This review delves into the potential of these innovative formulations to revolutionize the treatment of skin malignancies, due to their unique properties, having relevant advantages, such as allowing high drug strength, skin drug permeation and retention enhancement, biocompatibility, and controlled release capacity.
Despite the skin’s formidable permeability challenges, it remains an accessible interface for the delivery of therapeutic carriers such as nanoemulsions both locally (topical and dermal) and systemically (transdermal). Nanoemulsions, once associated primarily with cosmetic applications, are now gaining prominence as essential components of skin cancer treatment strategies. This review explores the potential of topical nanoemulsions, shedding light on their ability to efficiently deliver a wide range of molecules, overcoming lipophilic barriers inherent to skin. In this comprehensive analysis of several distinct studies investigating NEs for skin cancer treatment, a diverse array of formulations and components were explored, revealing a spectrum of characteristics.
Highlights
- Current skin cancer treatments often lack efficacy and lead to severe side effects.
- Topical nanoemulsions are powerful tools for skin cancer treatment.
- Nanoemulsions allow for increased skin drug delivery patterns and biocompatibility.
- Higher bioavailability and therapeutic efficacy are achieved using nanoemulsions.
- Nanoemulsions are promising topical drug delivery systems for skin cancer therapy.
The PDI spans from a minimum of 0.105 nm to a maximum of 0.421 nm, reflecting variations in droplet size distribution. Droplet sizes exhibit considerable diversity, ranging from a small 16 nm to a larger 200 nm, signifying varied potential for skin penetration. ZP values further contribute to this diversity, ranging from highly favorable (-66.6 mV) to less advantageous or near zero values, indicative of distinct surface charge characteristics.
As healthcare costs continue to escalate, this nuanced overview of nanoemulsion characteristics provides valuable insights into their potential applications in the targeted treatment of melanoma and, to a lesser extent, non-melanoma skin cancers. The value of such innovative and safer drug delivery systems becomes increasingly evident. Here, we focus exclusively on the role of topical nanoemulsions in advancing skin cancer therapy.
Table 2 Characteristics of the nanoemulsions utilized in melanoma and non-melanoma skin cancers management.
| NE | Materials | Skin cancer | Preparation technique | Droplet size (nm) | PDI | ZP (mV) | Key findings |
|---|---|---|---|---|---|---|---|
| Pund et. al (2015) – LFD-NEG | Transcutol® HP Capryol® 90 Cremophor® EL Pluronic® F127 | Melanoma | Self-emulsification | 123.7 | 0.278 | -7.8 | Halts tumor growth and suppresses cancer cell migration Specific and accelerated mortality rate in cell lines treated with the LFD-NE |
| Ugur Kaplan et al. (2019) – DZ-NEG | Ethyl oleate Lipoid S100 Tween® 80 DMSO Ultrapure water Protasan ™ UP G 213 | Melanoma | High energy homogenization | 149.8 | 0.222 | -19.32 | Enhanced viscosity for easier application and improved skin spreadability Maintained cytotoxic effectiveness against the melanoma cell line |
| Shakeel et al. (2015) 5-FU-NE | Lauroglycol-90 Deionized water Transcutol® HP Isopropyl alcohol | Melanoma | Spontaneous emulsification | 68.20 ± 2.65 | 0.219 | -25.92 | Transdermal chemoprevention attributes Significant drug permeation improvement |
| Kumar et al. (2015) 5-FU-NE | Span® 80 Span® 85 Tween® 80 Isopropyl alcohol Oleic acid Isopropyl myristate Triacetin | Non-melanoma | Oil phase titration | 100 | n.r. | n.r. | Higher skin retention and better control over the drug release in comparison to a topical 5-FU marketed cream |
| Tagne et al. (2008) DAC-NE | Ethanol Soybean oil Polysorbate 80 Ultrapure water | Melanoma | Microfluidization | 131 | 0.421 | − 5.49 | 10-fold greater tumor reduction Promising alternative to the conventional IV route |
| Dehelean et al. (2011) BA-NE | Flax-seed oil Egg phosphatidylcholine Deionized water | Melanoma | Microfluidization | 145 ± 1.5 | 0.4 | -39.1 ± 1.2 | Duality as prophylactic and therapeutic agent Interference with the angiogenic process |
| Chen et al. (2004) TPL-NE | Oleic acid Propylene glycol Tween® 80 1 % (v/v) menthol | n.r. | Magnetic stirring | Between 10 and 150 | Narrow | n.r. | Controlled, sustained, and prolonged delivery of TPL transdermally |
| Asasutjarit et al. (2021) AG-NE | Coconut, sesame, and jojoba oils Tween® 80 Lecithin Propylene glycol Ethanol Paraben concentrate | Melanoma and non-melanoma | Micro fluidization | 176.6 ± 1.8 | 0.332 ± 0.004 | − 11.78 ± 0.11 | Selective inhibitory activity against melanoma and non-melanoma cell lines Convenient reduced dosing frequency |
| Liu et al. (2021) CNE | Soybean oil Vitamin E, Tween® 80 Lecithin Deionized water | Melanoma | Homogenization and ultrasonication | 16 | 0.132 | − 66.6 | Dose-dependent decrease in cell viability |
| Nasr et al. (2022) TEO-NE | TEO Lecithin Tween®80 Amphiphilic oligochitosan | Melanoma | Low-energy emulsification | 184.74 ± 1.27 | 0.19 | +23.82 ± 0.55 | Bilayered emulsion modified with an oppositely charged biopolymer Strong attachment to negatively charged PS expressed on the melanoma cells surface |
| Nagaraja et al. (2021) Chrysin-NE | Caproyl® 90 Tween® 80 Transcutol® HP | Melanoma | Self-emulsification | 156.9 ± 3.4 | 0.26 | -15 | Transforming chrysin into a nanoemulgel formulation augmented its therapeutic efficacy |
| Shakeel et al. (2010) CAF-NE | Lauroglycol-90 Transcutol® HP Isopropyl acid | Skin cancer caused by sun exposure | Oil phase titration | 20.14–105.25 | 0.105–0.177 | n.r. | NE components functioned as permeation enhancers, obviating the need for additional chemicals Safe and effective transdermal delivery |
| Tang et al. (2021) THC-NE | Transcutol® Medium-chain triglycerides | Melanoma | High-speed and highpressure homogenization | n.r. | n.r. | n.r. | Protective effects from H2O2- induced cell death Effective inhibition of α-MSH induced melanin production |
| Guerrero et al. (2018) CUR-NE | Miglyol 812 Epikuron 145V Ethanol Acetone Ultrapure water | Melanoma postsurgery reincidence and metastasis | Spontaneousemulsification | 200 | ≤ 0.2 | -30 | Specific cytotoxicity against cancer cell lines, including melanoma B16F10 cells Prevention of tumor recurrence and spontaneous lung metastasis |
| Dalmolin et al. (2018) ZnPc-NE | MCTs DOTAP Lipoid E80 Water Polysorbate 80 Poloxamer 188 | Melanoma | Ultrasonication | 200 | 0.20 ± 0.02 | 43 ± 7 | Enhanced ZnPc skin permeation through synergistic nanocarrier and iontophoresis Improved ZnPc penetration and uniform drug distribution within the tumor |
| Martínez-Razo et al. (2023) NCTD-NE | Almond oil Urea Glyceryl monostearate Glycerin Cetyl alcohol Stearic acid Polysorbate 80 Eumulgin B1® | Melanoma | Ultrasonication | 117 to 120 | 0.26 to 0.28 | 0 | Consistent prolonged NCTD release Approximately 10-fold solubility augment |
Download the full article as PDF here Topical delivery of nanoemulsions for skin cancer treatment
or read it here
Joana Duarte, Ankur Sharma, Esmaeel Sharifi, Fouad Damiri, Mohammed Berrada, Moonis Ali Khan, Sachin Kumar Singh, Kamal Dua, Francisco Veiga, Filipa Mascarenhas-Melo, Patrícia C. Pires, Ana Cláudia Paiva-Santos,
Topical delivery of nanoemulsions for skin cancer treatment, Applied Materials Today, Volume 35, 2023, 102001, ISSN 2352-9407, https://doi.org/10.1016/j.apmt.2023.102001.

