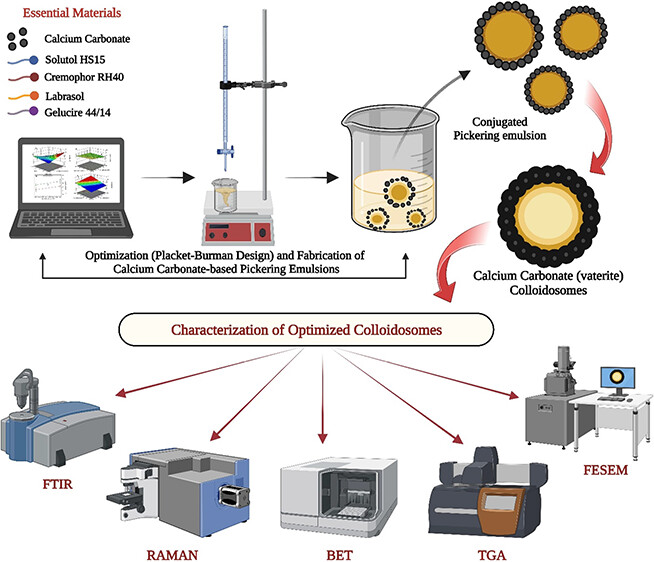
The objective of the research was to identify significant variables that impact the porosity-related properties of CaCO3 particles. The Placket–Burman design was employed to screen multiple variables, including pH, molar concentrations of calcium chloride and sodium carbonate, temperature, concentration of Gelucire 44/14, Cremophor RH40, Solutol HS15, Labrasol, mixing rate, reaction time, and order of addition. The response variables were surface area, pore radius, and pore volume. Influential methodologies such as XRD, FTIR, Raman spectroscopy, and TGA were utilized to validate the precipitate type. The BET surface area ranged from 1.5 to 16.14 m2/g, while the pore radius varied from 2.62 to 6.68 nm, and the pore volume exhibited a range of 2.43 to 37.97 cc/gm. Vaterite structures with spherical mesoporous characteristics were observed at high pH, whereas calcite formations occurred at low pH. The order of addition impacted the surface area but did not affect the pore volume. To maximize the surface area, a lower reaction time and molar concentrations of sodium carbonate were found to be advantageous. The pore radius was influenced by the pH, surfactants, and reaction conditions. The sediments were categorized based on the percentage of vaterite formation. The instrumental techniques effectively characterized the precipitates and provided a valuable complementary analysis.
1. Introduction
Porous vaterite calcium carbonate (CaCO
3), owing to large porosity, high surface area, and rapid decomposition in acidic conditions, contributes to a practical and alternate drug delivery choice/sacrificial template to ferry drugs. (1,2) Vaterite CaCO
3 has been reported as a host for various moieties such as doxorubicin, (3)
antimicrobials, (4) rhodamine B, (5) photosens, (6) and methotrexate, (7) because of the inherently high surface area. The occurrence of vaterite in nature is relatively rare, primarily due to thermodynamic constraints that hinder the direct transformation of calcite into vaterite. (8) Among various synthetic approaches (such as biomimetic synthesis and CO
2-bubbling method) for preparing vaterite particles, the precipitation method using precursor salts (Na
2CO
3 and CaCl
2) is relatively simple and industrially feasible. (8,9) Mixing of the precursor salt seems to be an attractive alternative for the preparation of porous vaterite; however, the number of independent variables such as pH, temperature, reaction time, mixing mode, additives (surfactants, polymers, biomolecules, amino acids), and solvent ratio needs to be aptly controlled and optimized for its preparation. In the present research, we aim to screen and evaluate the critical independent variables (InV) influencing the response variables related to porosity such as surface area, pore radius, and pore volume. To achieve this, we employed the Placket–Burman design (PBD). The selected InV values were as follows: (a) pH, (b) molar concentration of calcium chloride, (c) molar concentration of sodium carbonate, (d) temperature, (e) concentration of additives (Gelucire 44/14, Cremophor RH40, Solutol HS15, Labrasol), (f) mixing rate, (g) reaction time, and (h) order of addition (L1, L2). Additives (Gelucire 44/14, Cremophor RH40, Solutol HS15, Labrasol) were selected based on our previous report that gave a maximum mole fraction of vaterite compared to calcite and aragonite.(5)
Considering the apparent relationship between drug loading, surface area, pore radius, and pore volume were selected as response variables (ReV). Among various screening designs, we have chosen the PBD because it can identify the main factors from a pool of many InV (as in the present work) that have the most impact on the selected responses. (10) During our literature review on preparing porous vaterite CaCO3, we identified a gap in knowledge regarding the relationship between different variables and their impact on the surface area, pore radius, and pore volume. These properties are crucial in determining the drug loading capacity and release behavior of the bioactive substances. To address this gap, the present research focuses on screening and evaluating the variables influencing response (surface area, pore radius, and pore volume) to enhance our understanding of this field. In one of our previous studies, we studied the impact of eleven different excipients on the phase transition behaviors of calcite, vaterite, and aragonite. (5) To ascertain the authenticity of the precipitates formed through the PBD, we validated them by employing X-ray diffraction (XRD), Fourier transform infrared (FTIR) spectroscopy, Raman spectroscopy (RS), and thermogravimetric analysis (TGA). Also, we have classified the precipitates into three distinct groups based on vaterite content. Group 1 consists of sediments with a vaterite percentage exceeding 75%; Group 2 includes residues with a vaterite content ranging from 45% to 70%; and Group 3 exclusively comprises calcite forms. This classification provides valuable insights into the varying compositions of the residues and facilitates a more comprehensive analysis of the experimental results.
2.1. Materials
Calcium chloride (CaCl2) and sodium carbonate (Na2CO3) were acquired from Sigma Aldrich (India) for this study. Additionally, Solutol HS15 and Cremophor RH40 were generously provided as gift samples by BASF (USA), while Labrasol and Gelucire 44/14 were supplied ex gratia by Gattefosse (France). A mechanical stirrer (IKA RW 20 digital, India) was used to mix the contents. GPT-4 and Grammarly served as AI-assistants in drafting the final manuscript.
Download the full study as PDF here: Characterization and Exploration of Placket–Burman-Designed Porous Calcium Carbonate (Vaterite) Microparticles
or read it here
Avi Singh, Sabya Sachi Das, Priya Ranjan Prasad Verma, Janne Ruokolainen, Kavindra Kumar Kesari, and Sandeep Kumar Singh. Characterization and Exploration of Placket–Burman-Designed Porous Calcium Carbonate (Vaterite) Microparticles. ACS Omega Article ASAP.









































 All4Nutra
All4Nutra







