StarLac – MEGGLE’s co-processed lactose grades for direct compression

StarLac - Technical brochure by MEGGLE
General Information
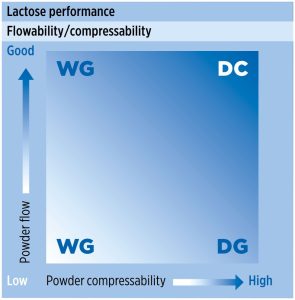
Direct compression (DC) tablet manufacture is a popular choice because it provides the least complex, most cost effective process to produce tablets compared to other tablet manufacturing approaches. Manufacturers can blend APIs with excipients and compress, making dosage forms simple to produce [1, 2].
DC technology and the use of modern tableting equipment require that excipients and APIs form a compactable mixture with excellent flowability and low particle segregation tendency [3].
In the pharmaceutical industry, lactose is one of the most commonly used excipients; however, like many other excipients, lactose may not be suitable for direct compression without modification due to insufficient powder flow or/and compaction properties (figure 1).
Product description
Alpha-lactose monohydrate and maize starch (corn starch) are functional excipients used in oral solid dosage forms. Both are naturally derived and well established in the pharmaceutical industry. Lactose is frequently used as a diluent or direct compression binder. Starch can be used as a binder for wet or dry applications, disintegrant, and diluent. In an effort to establish synergistic functional performance, such as enhanced compactibility and faster tablet disintegration, lactose and starch were co-spray-dried to form a monoparticulate system. StarLac® comprises 85 % alpha-lactose monohydrate and 15% native maize starch. StarLac® provides compaction and lubricant insensitivity characteristics desired for direct compression, and the hydration properties desired for rapid API release. Additionally, StarLac®’s flowability is superior compared to a physical blend of the individual components in equivalent ratio.
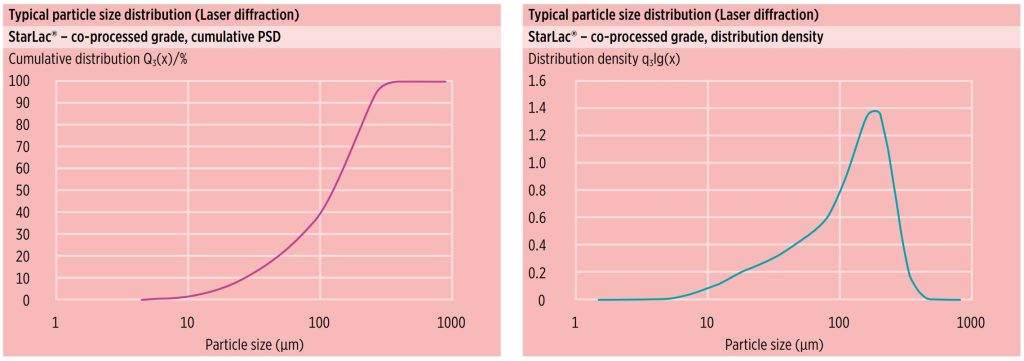
Particle size distribution (PSD)
Figure 2 shows typical laser diffraction particle size distribution data for StarLac®. StarLac® possesses a narrow PSD that supports homogenous powder blend preparation, essential for achieving good tablet quality.
Figure 3 depicts the specified PSD range and typical average values by air-jet sieving. These parameters are constantly monitored through in-process control (IPC) testing and are part of the StarLac® particle size distribution specification.
Functional related characteristics
Powder flow
In assessing powder flow using a FlowRatex® apparatus, StarLac® exhibited superior flowability compared to a physical blend made up of spray-dried lactose and maize starch. The simple blend of individual ingredients showed greater flow variation compared to StarLac® (figure 7). StarLac® also possessed lower flowability index (StarLac® = 2 mm, physical blend = 16 mm), indicating superior flowability (figure 8).
See the full brochure on “StarLac®” here
(click the picture to download the brochure)
Benefits StarLac®
- Excellent compactibility
- Excellent flowability
- Rapid, hardness-independent tablet disintegration
- Compaction and hydration properties do not dendent on hydrophobic lubricant type or leve
Source: MEGGLE brochure “StarLac®”
See the overview video of the MEGGLE Dry Powder Inhalation product range here:
Company: MEGGLE is one of the world´s leading manufacturers of pharmaceutical grade lactose and co-processed excipients with expertise of more than 70 years. We encounter lactose in so many areas of our life – reason enough to take a closer look at this multi-functional “white powder”.
Do you need more information or a sample of StarLac® excipients?









































 All4Nutra
All4Nutra








