Novel SmartReservoirs for hydrogel-forming microneedles to improve the transdermal delivery of rifampicin
Novel SmartReservoirs for hydrogel-forming microneedles to improve the transdermal delivery of rifampicin
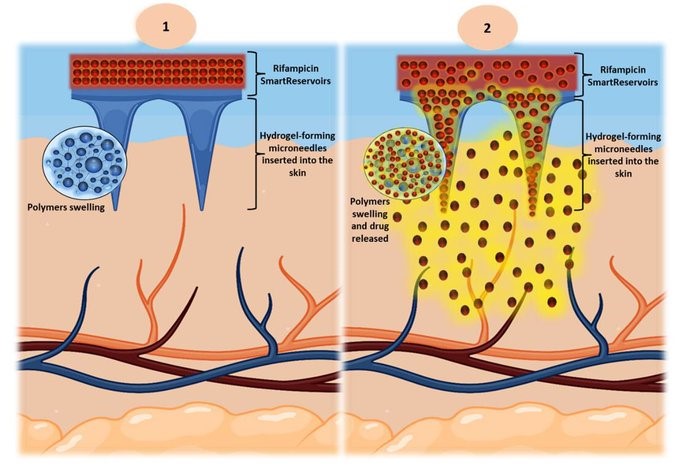
Hydrogel-forming microneedles (HF-MNs) are composed of unique cross-linked polymers that are devoid of the active pharmaceutical ingredient (API) within the microneedle array. Instead, the API is housed in a reservoir affixed on the top of the baseplate of the HF-MNs. To date, various types of drug-reservoirs and multiple solubility-enhancing approaches have been employed to deliver hydrophobic molecules combined with HF-MNs. These strategies are not without drawbacks, as they require multiple manufacturing steps, from solubility enhancement to reservoir production. However, this current study challenges this trend and focuses on the delivery of the hydrophobic antibiotic rifampicin using SmartFilm-technology as a solubility-enhancing strategy.
In contrast to previous techniques, smart drug-reservoirs (SmartReservoirs) for hydrophobic compounds can be manufactured using a one step process. In this study, HF-MNs and three different concentrations of rifampicin SmartFilms (SFs) were produced. Following this, both HF-MNs and SFs were fully characterised regarding their physicochemical and mechanical properties, morphology, Raman surface mapping, the interaction with the cellulose matrix and maintenance of the loaded drug in the amorphous form. In addition, their drug loading and transdermal permeation efficacy were studied. The resulting SFs showed that the API was intact inside the cellulose matrix within the SFs, with the majority of the drug in the amorphous state. SFs alone demonstrated no transdermal penetration and less than 20 ± 4 μg of rifampicin deposited in the skin layers.
In contrast, the transdermal permeation profile using SFs combined with HF-MNs (i.e. SmartReservoirs) demonstrated a 4-fold increase in rifampicin deposition (80 ± 7 μg) in the skin layers and a permeation of approx. 500 ± 22 μg. Results therefore illustrate that SFs can be viewed as novel drug-reservoirs (i.e. SmartReservoirs) for HF-MNs, achieving highly efficient loading and diffusion properties through the hydrogel matrix.
Download the full article as PDF here: Novel SmartReservoirs for hydrogel-forming microneedles to improve the transdermal delivery of rifampicin
or read it here
Materials
Rifampicin was purchased from Alfa Aesar (Lancashire, UK). Methanol 96% (v/v), Poly(vinyl alcohol) with molecular weight of 85–124 kDa, citric acid, orthophosphoric acid, sodium lauryl sulphate and ascorbic acid were purchased from Sigma-Aldrich (Dorset, UK). Poly(vinyl pyrrolidone) K29-32 (PVP) with molecular weight of 58 kDa was provided by Ashland (Kidderminster, UK). Ultrapure water was obtained from a water purification system (HPLC grade, Elga PURELAB DV 25, Veolia Water Systems, Dublin, Ireland).
Abraham M. Abraham, Qonita Kurnia Anjani, Masoud Adhami, Aaron R. J. Hutton, Eneko Larran˜eta and Ryan F. Donnelly, Novel SmartReservoirs for hydrogel-forming microneedles to improve the transdermal delivery of rifampicin, Received 16th January 2024, Accepted 7th March 2024, DOI: 10.1039/d4tb00110a, Cite this: DOI: 10.1039/d4tb00110a, Journal of Materials Chemistry B, rsc.li/materials-b
Read more articles on Rifampicin here:
- 3D printed hollow microneedles array using stereolithography for efficient transdermal delivery of rifampicin
- Rifampicin nanocrystals: Towards an innovative approach to treat tuberculosis
- Development of porous particles using dextran as an excipient for enhanced deep lung delivery of rifampicin










































 All4Nutra
All4Nutra







