Enhancing Drug Efficacy: Gattefossé’s Expertise in Excipient Selection

The Gattefossé Group is a community of employees, all driven by a dual mission: the performance of its products and the personalized support to its customers. Gattefossé develops, manufactures and sells cosmetic ingredients and pharmaceutical excipients of natural origin for the beauty and health industries worldwide, through its 12 affiliated companies and network of global agents and distributors in 80 countries.
Environmental and social issues have always been part of the culture of this French and independent family business, founded in Lyon in 1880. Today, Gattefossé relies on a purposeful Corporate Social Responsibility (CSR) approach to build its innovation and development strategy.
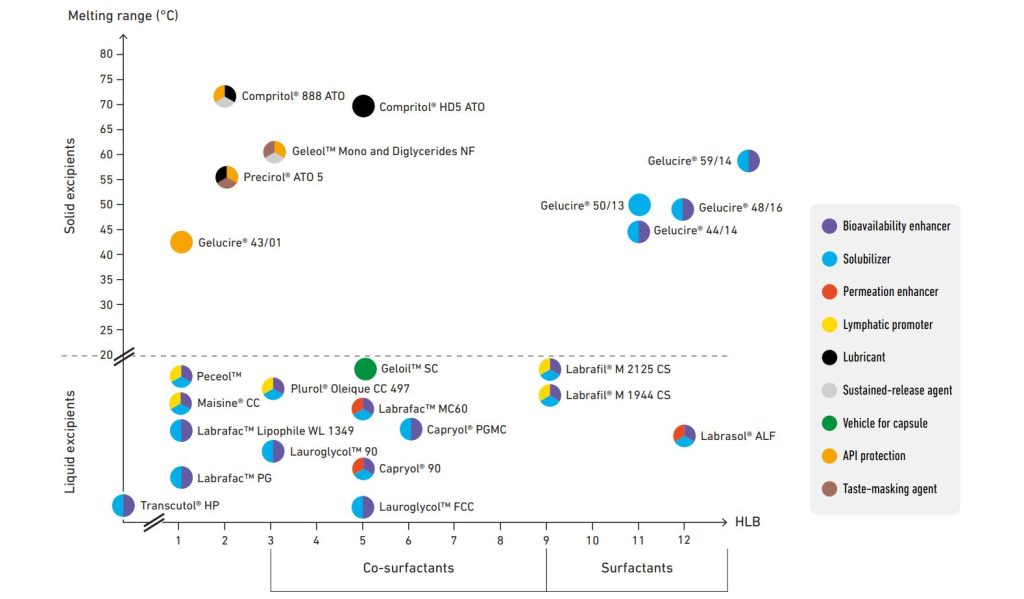
Oral drug delivery
Enhancing oral bioavailability
Poor solubility, poor permeability, and pre-systemic elimination are factors that can limit absorption of drugs. Our excipients have the capability to enhance oral bioavailability of small molecules, peptides and macromolecules by:
- Maintaining drug in a solubilized state throughout the gastro-intestinal tract
- Transiently increasing intestinal permeation
- Promoting lymphatic uptake
Our range of functional excipients includes oils, co-surfactants, surfactants and solvents. They are easily combined to formulate all types of lipid-based formulations and self-emulsifying drug delivery systems.
Sustaining drug release
The development of lipid sustained-release matrices is straightforward and provides the following biopharmaceutical and manufacturing advantages:
- Effective modulation of drug release profile for highly water soluble, short half-life drugs
- Reduced risk of alcohol dose dumping and misuses
- Non-hygroscopic matrix for improved stability on storage
- Solvent-free processing for cleaner and greener manufacturing
- Suitable for cold and hot manufacturing processes: direct compression, granulation, 3D printing, hot melt extrusion
Lubricating tablets and capsules
Effective lubrication in tablet and capsule production has a major impact on the manufacture and the quality of the final dosage forms. Our lubricants provide valuable formulation and process advantages:
- Chemical inertness, for high compatibility with a wide range of molecules
- Insensitive to mixing conditions, for highly robust process and formulation
- Efficient, while maintaining tablet properties such as hardness and drug release
Taste masking and API protection
A continuous lipid film coating around the drug particles or tablets enables to isolate the bad tasting drug from the taste buds, or to protect the sensitive drug from environment conditions such as light, moisture, oxidation or temperature, and prevent its degradation.
Lipid excipients can be processed with a variety of techniques, such as high shear coating, hot melt coating, fluid bed coating or melt granulation to provide a robust lipid film coating.
Skin drug delivery
Enhancing skin delivery
Our excipients solubilize a wide range of molecules. Lipid excipients have amphiphilic properties, and can modulate the penetration of drugs into the stratum corneum and drive its flux.
Our penetration enhancers have different modes of action involving interactions at different levels with the skin:
- Interaction with the protein or lipid domains
- Transiently opening of the tight junctions between cells
- Membrane fluidization
A combination of penetration enhancers with different mechanisms provides interesting synergies and contribute to the push and pull effect, to optimize topical or transdermal drug delivery.
Developing patient-friendly textures
Our excipients enable the formulation of all topical and transdermal dosage forms, with attractive textures aiming at improving patient adherence, while ensuring safe and efficient topical or transdermal drug delivery. Our range includes solvents, penetration enhancers, emulsifiers, co-emulsifiers, surfactants, co-surfactants, thickeners, emollients and oily vehicles.
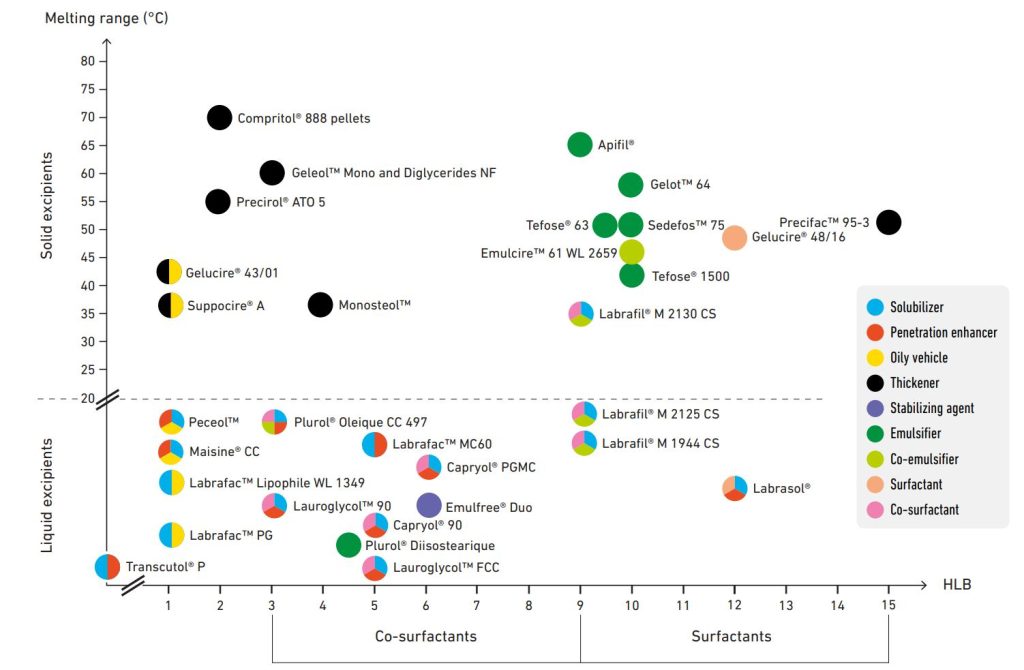
Solvent – Penetration enhancers
• Capryol® 90 *
• Capryol® PGMC *
• Labrafac™ MC60 *
• Labrasol®
• Lauroglycol™ 90 *
• Lauroglycol™ FCC *
• Maisine® CC *
• Peceol™ *
• Plurol® Oleique CC 497 *
• Transcutol® P *
Emulsifiers
• Apifil®
• Gelot™ 64
• Plurol® Diisostearique *
• Sedefos™ 75
• Tefose® 1500
• Tefose® 63
Surfactants
Co-emulsifiers
• Emulcire™ 61 WL 2659
• Labrafil® M 1944 CS
• Labrafil® M 2125 CS
• Labrafil® M 2130 CS
• Plurol® Oleique CC 497 *
Co-surfactants
• Capryol® 90 *
• Capryol® PGMC *
• Lauroglycol™ 90 *
• Lauroglycol™ FCC *
• Labrafil® M 1944 CS
• Labrafil® M 2125 CS
• Labrafil® M 2130 CS
• Plurol® Oleique CC 497 *
Stabilizing agent – Thickeners
• Emulfree® Duo *
• Compritol® 888 Pellets *
• Geleol™ Mono and Diglycerides NF *
• Gelucire® 43/01 *
• Monosteol™ *
• Precifac™ 95-3 *
• Precirol® ATO 5 *
Oily vehicles – Emollients
• Gelucire® 43/01 *
• Labrafac™ PG *
• Labrafac™ Lipophile WL 1349 *
• Peceol™ *
• Suppocire® A *
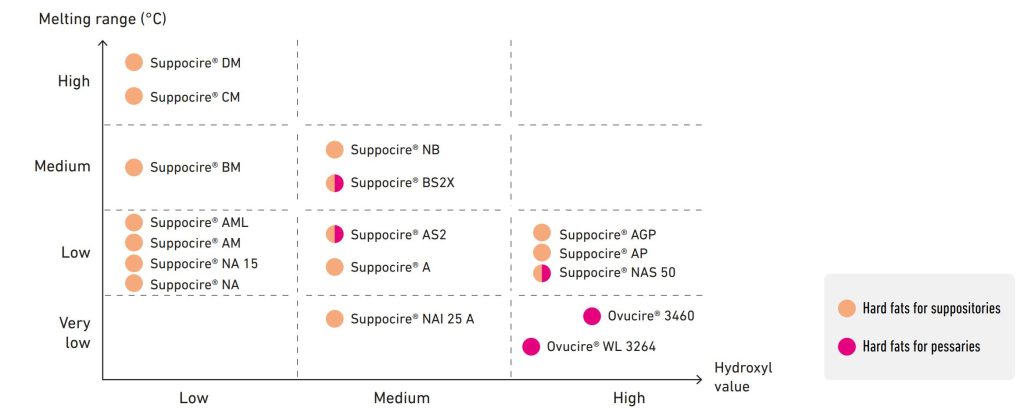
Rectal and vaginal drug delivery
Suppositories and pessaries
Our hard fat bases Suppocire® and Ovucire® provide:
- Proven safety and mucosal tolerance
- Excellent drug dispersion and physico-chemical stability
- Narrow melting range for high performance in vivo
- Solidification behavior adapted to a wide range of manufacturing equipments
Our bases and formulation expertise enable to:
- Simplify existing formulations by reducing the number of excipients
- Optimize the manufacturing process and reduce quality issues
- Obtain excellent product quality attributes including drug dissolution, i.e. efficacy
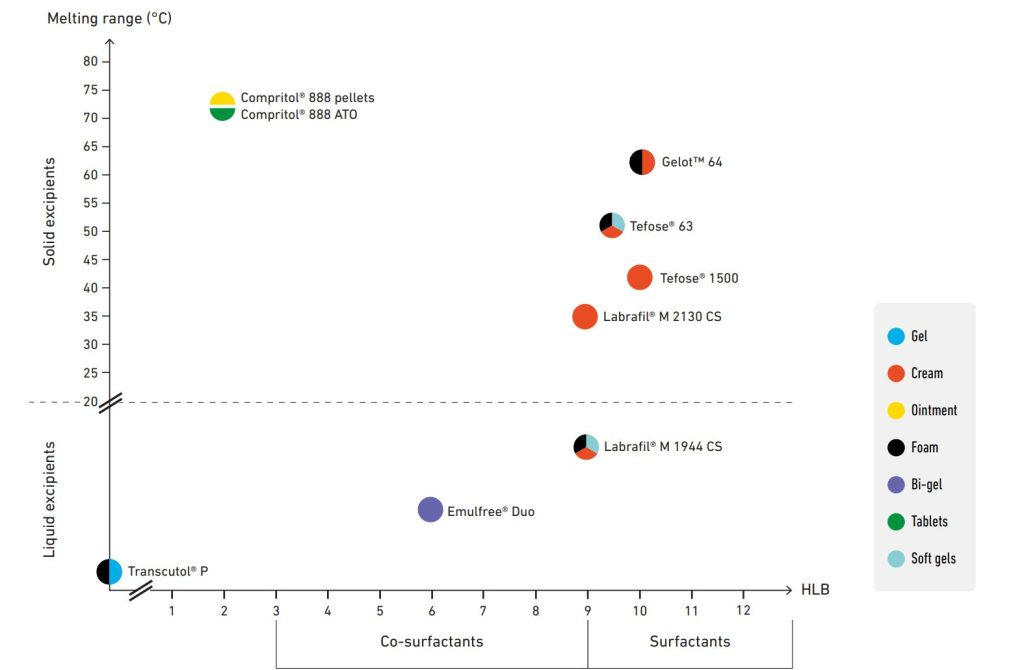
Alternative dosage forms
If solid dosage units, i.e. suppositories and pessaries, are very frequent for rectal and vaginal drug delivery, other dosage forms for local application are also used: creams, gels, bi-gels, foams, ointments, tablets… to achieve a local
or systemic effect.
See the full technical brochure on “Optimize drug delivery” here
(click the picture to download the brochure)
Source: Gattefossé technical brochure “Optimize drug delivery”
Do you need more information or a sample of Gattefossé excipients?


